CHMB42H3 Lecture Notes - Lecture 14: Wittig Reaction, Triphenylphosphine, Sodium Cyanoborohydride
Document Summary
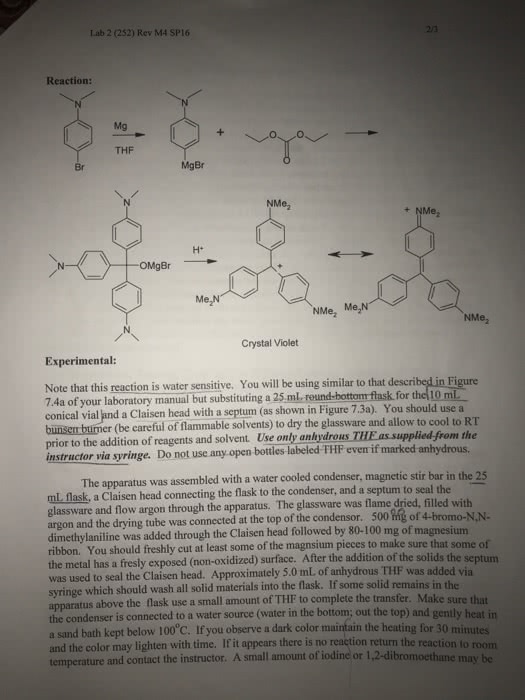
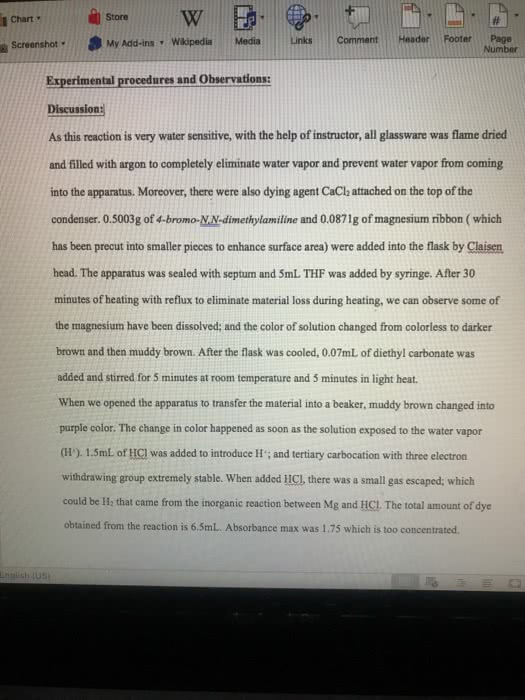
Chapter 17 reactions of aldehydes & ketones/reactions of carboxy. Derivatives/reactions of , -unsaturated carbonyl compounds alkyl groups stabilize the carbonyl compound & destabilize the transition state. Irreversible b/c two en attached to the ti (whichever can be protonated) & eliminated. 17. 4 the reactions of carbonyl compounds wit grignard reagents. Alkoxide ion is stable because it does not have a group that can be eliminated. Forms primary (formaldehyde) and secondary and tertiary (ketone) alcohols. If the reaction w/ the carbonyl compound forms a product with an asymmetric center the product will be a racemic mixture. But if it is catalyzed by an enzyme, only one of the enantiomers is formed. The reactions of ester and acyl chlorides w/ grignard reagents. The first reaction is nucleophilic addition-elimination b/c an ester or an acyl chloride has a group that can be replaced by the grignard reagent. The product of an ester w/ grignard is a tertiary alcohol.