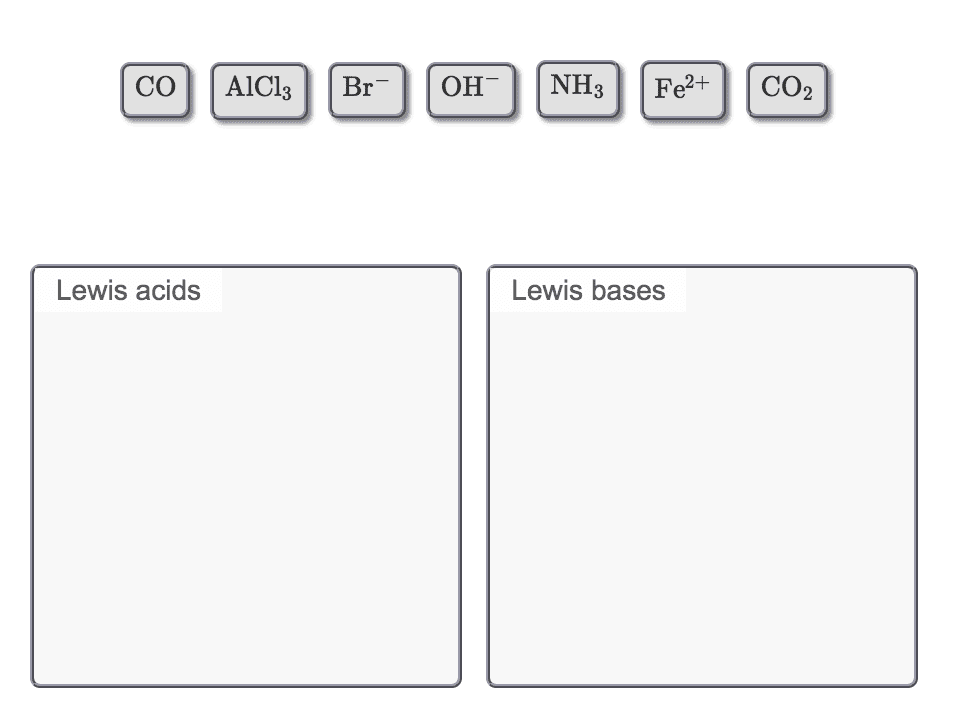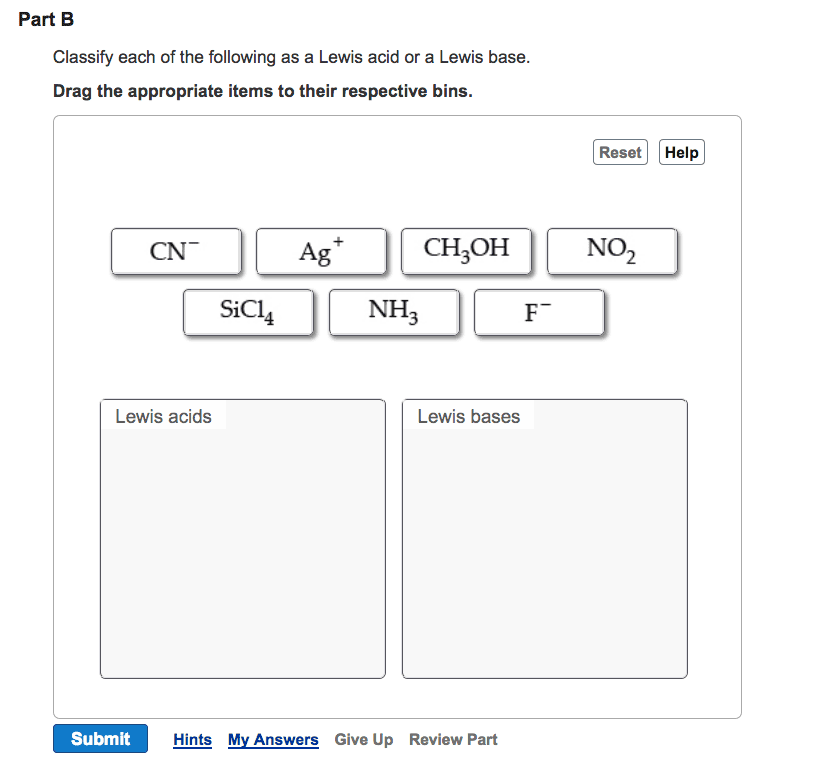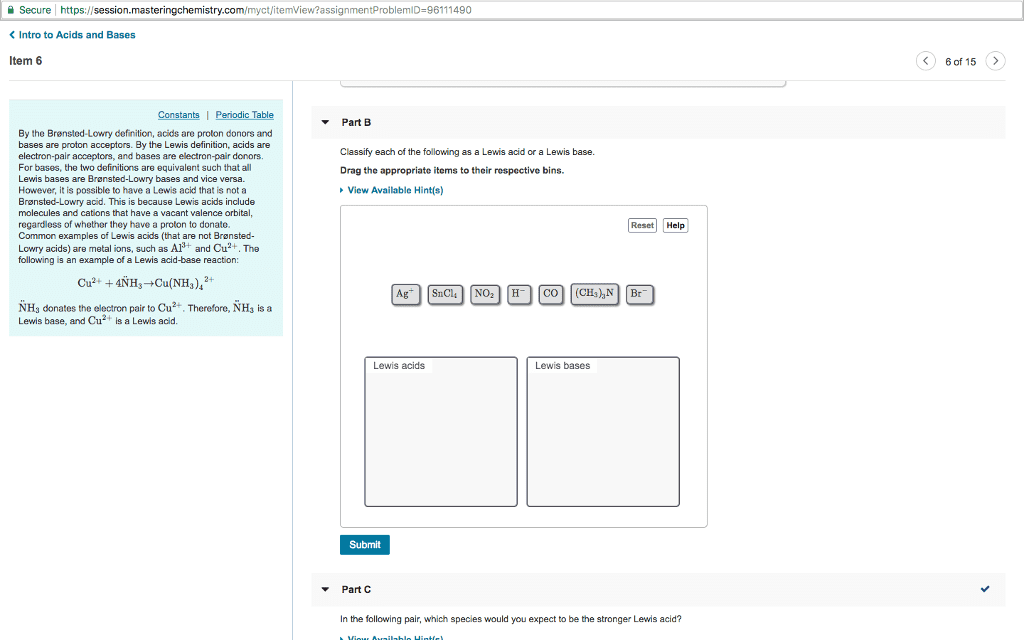
ì Secure! https://session.masteringchemistry.com/myct/itemview?assignmentProblemiD-96111490 Intro to Acids and Bases Item 6 6 of 15> â¼ Part B By the Brønsted-Lowry definition, acids are proton donors and bases are proton acceptors. By the Lewis definition, acids are electron-pair acceptors, and bases are electron-pair donors. For bases, the two definitions are equivalent such that al Lewis bases are Bronsted-Lowry bases and vice versa. However, it is possible to have a Lewis acid that is not a Brønsted-Lowry acid. This is because Lewis acids include molecules and cations that have a vacant valence orbital, regardless of whether they have a proton to donate. Common examples of Lewis acids (that are not Bronsted- Lowry acids) are metal ions, such as A and Cu2+. The following is an example of a Lewis acid-base reaction: Classify each of the following as a Lewis acid or a Lewis base. Drag the appropriate items to their respective bins View Available Hint(s) Reset Help Cu2 +4NH+Cu(NH NHs donates the electron pair to Cu2 Therefore, NH3 is a Lewis base, and Cu2 is a Lewis acid. Lewis acids Lewis bases Submit Part C In the following pair, which species would you expect to be the stronger Lewis acid?