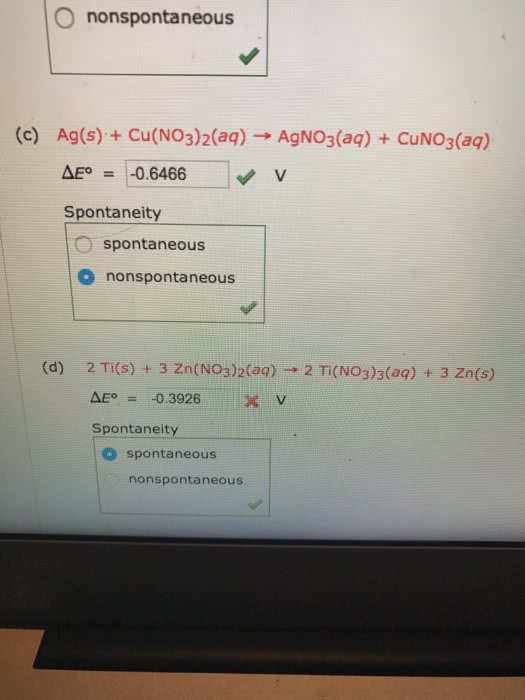
CHM135H1 Lecture : Spontaneity and E^0 Cell (Easy)
29 views2 pages
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related Documents
Related Questions
At constant temperature and pressure, which statement gives the relationship between the sign of ÎG and the spontaneity of the reaction?
| 1) When ÎG < 0, the forward reaction is spontaneous. | |
| 2) When ÎG > 0, the forward reaction is spontaneous. | |
| 3) When ÎG < 0, the reverse reaction is spontaneous. | |
| 4) When ÎG = 0, the forward reaction is spontaneous. |
Can someone please help