Calculate Delta G rxn for the four reactions in blue.
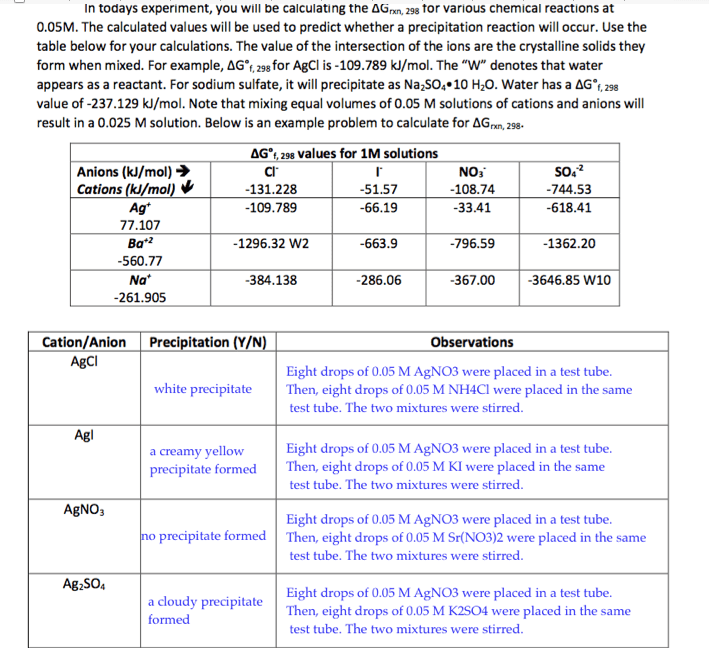
In todays experiment, you will be calculating the AG ron, 298 tor various chemical reactions at 0.05M. The calculated values will be used to predict whether a precipitation reaction will occur. Use the table below for your calculations. The value of the intersection of the ions are the crystalline solids they form when mixed. For example, AG 298 for AgCl is -109.789 kJ/mol. The "W" denotes that water appears as a reactant. For sodium sulfate, it will precipitate as Na2SO4.10 H20. Water has a AG fi, 298 value of -237.129 kJ/mol. Note that mixing equal volumes of 0.05 M solutions of cations and anions will result in a 0.025 Msolution. Below is an example problem to calculate for AGran 298- AG. 298 values for 1M solutions Anions (kJ/mol) NO3 Cations (k/mol) -131.228 -744.53 -109.789 -33.41 77.107 Ba 2 -796.59 -1362.20 -1296.32 W2 -3646.85 W10 -384.138 -261.905 Cation/Anion Precipitation (Y/N) Observations AgCl Eight drops of 0.05 M AgNO3 were placed in a test tube white precipitate Then, eight drops of 0.05 M NH4Cl were placed in the same test tube. The two mixtures were stirred. a creamy yellow Eight drops of 0.05 M AgNO3 were placed in a test tube precipitate formed Then, eight drops of 0.05 M KI were placed in the same test tube. The two mixtures were stirred. AgNO, Eight drops of 0.05 M AgNO3 were placed in a test tube no precipitate formed Then, eight drops of 0.05 M Sr(NO302 were placed in the same test tube. The two mixtures were stirred. Ag2SO4 Eight drops of 0.05 M AgNO3 were placed in a test tube a cloudy precipitate Then, eight drops of 0.05 M K2SO4 were placed in the same formed test tube. The two mixtures were stirred