Question 2 Energy Calculations
a) Calculate the heat energy (q) when 12.0 g of water is heated from 20oC to 100oC. The
specific heat for water is given in your textbook.
b) Is the process of heating water exothermic or endothermic? Explain your answer.
Question 3 Energy Calculations
You dissolve 0.1 mol of ammonium nitrate in 100 mL of H2O. The temperature of the solution
changes from 22oC to 15oC. The specific heat capacity for water is 4.184 J/g.K. Show all
calculations on a separate sheet of paper!
a) Is this reaction endothermic or exothermic?
b) Calculate the heat, q, released or absorbed in the dissolution reaction.
c) Calculate the enthalpy change, ?Hrxn (kJ/mol), for dissolving 1 mol of ammonium nitrate.
Question 4. A gas expands from 264 mL to 971 mL at constant temperature. Calculate
the amount of work done in Joules by the gas when it expands against a constant pressure of 955
torr.
Question 5 Energy Calculations
a) The nutritional calorie (Calorie) is equivalent to 1 kcal. One pound (1.0 lb) of body fat is
equivalent to 4.1 x 103 kcal. Express this quantity in joules and kilojoules. Use
scientific notation for your answer!
b) If an athlete expends 1850 kJ/h (kilojoules per hour), how long does he have to exercise
to work off 1.0 lb of body fat?
c) The average amount of calories burned during running is 125 kcal/mile. How many
miles does this athlete have to run to burn off 1.0 lb of body fat?
Question 6
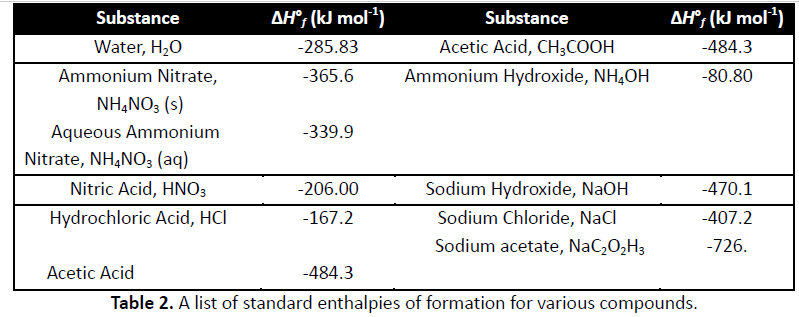
Calculate ?Ho for the following reactions at 298 K. Give the balanced thermochemical
equations and show the correct physical state in each equation. Calculate the enthalpy change
for each reaction. You will find thermochemical data in the back of your textbook in Appendix 3
(p. A-8 to A-12)
a) The formation of 1 mol of HCl (gas) from its elements
b) The formation of 1 mol of sodium chloride from its elements
c) The combustion of 1 mol glucose to water and carbon dioxide
d) HCl(g) + NH3(g) ? NH4Cl(g)
Question 7
Calculate heat of formation of SO2 under nonstandard state conditions, given the following
equations
SO2(g) +