CHM135H1 Lecture : Notes taken during lecture

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
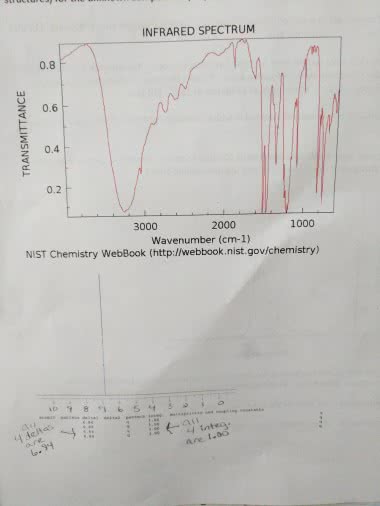
Notes a mole = 6. 022 x 10^23 units of something therefore sensible for calculations about atomic-scale entities. 1 mol = 12 g of ^12 c. Mass to moles via dividing by g/mol. Moles to number of atoms via multiplying by avogadro"s. 29 number number of atoms to moles via dividing by avogadro"s number. Moles to mass via multiplying by g/mol. Fill in the blank with the mws of the four options to determine d. Formula to determine number of molecules: significance of empirical formulas: when presented with unknown compound, one uses combustion analysis, which releases elemental gases present in a certain ratio (thus determining empirical formula) Mass % = mass of element per mole of compound x 100% 2 mol of o (15. 999g/mol) + 1 mol of m (x) x is the molar mass of m solve for x, which is 47. 90 g/mol -> titanium therefore, 40. 05% = 2 mol of o (15. 999g/mol) x 100%