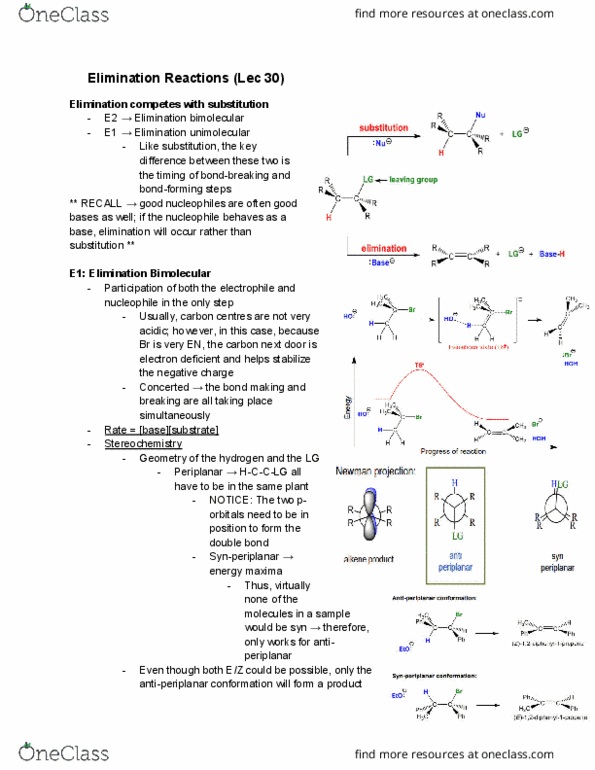
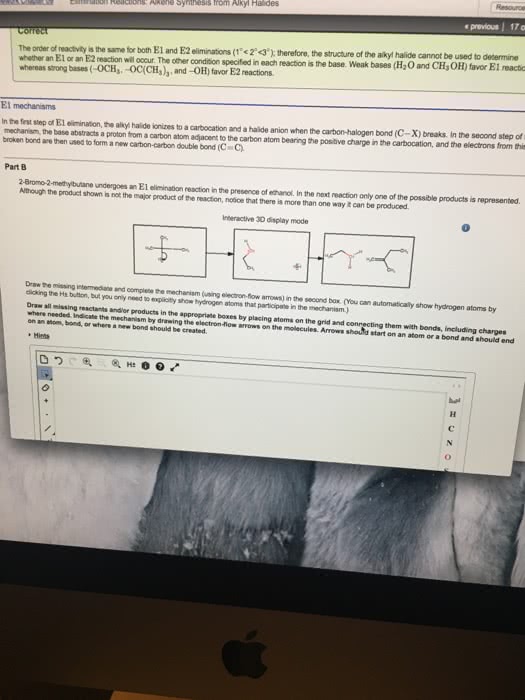
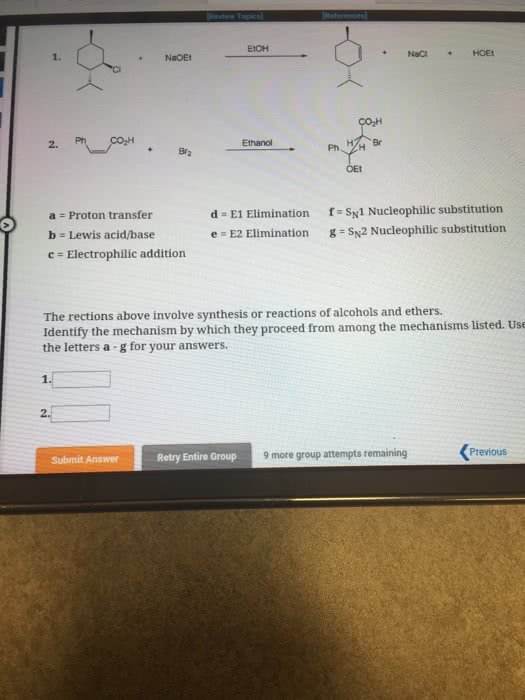
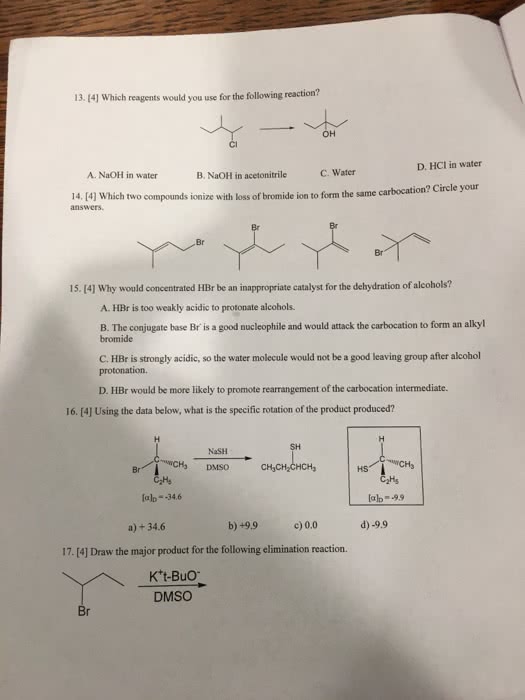
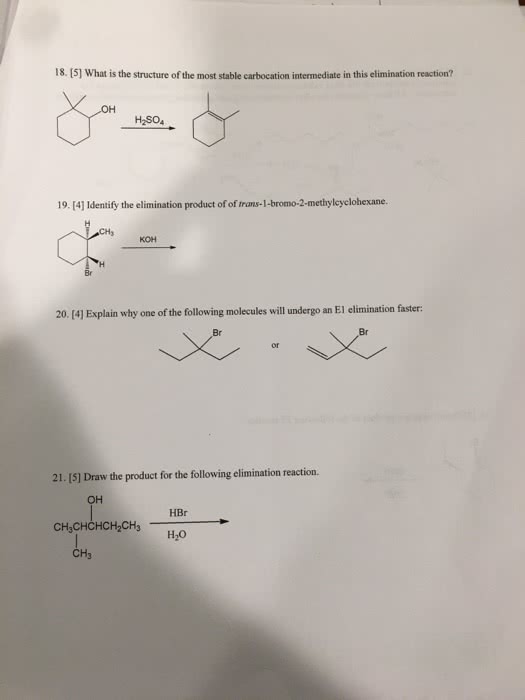
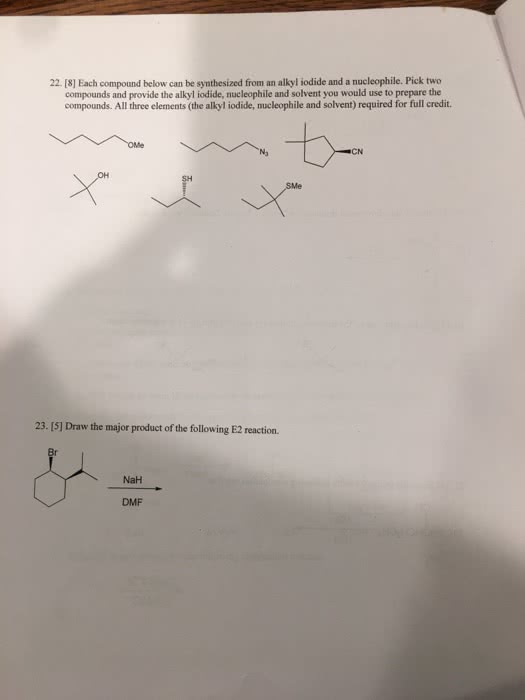
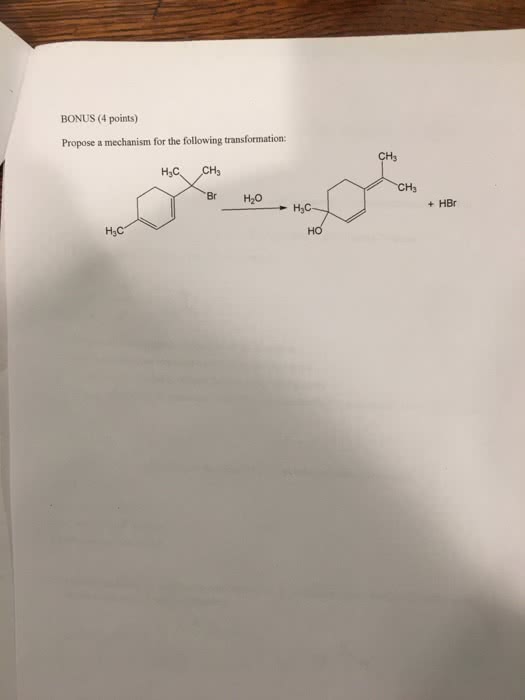
previous 17 a The order of reactivity is the same for both E1 and E2 eliminations (1 2therefore, the structure of the alk n wll ocaur The other condion specifed in each reaction is the base. Weak bases (H20 and CH OH) favor El reactic whereas strong bases(OCHs -OC(CHs),and-OH) favor E2 reactions. In the first step of E1 elimination, the alkyl halide ionizes to a carbocation and a halide anion when the carbon-halogen bond (C-X) breaks In the second step of from this mechanism, the base abstracts a proton from a carbon atom adjacent to the carbon atom bearing the positive charge in the carbocation, and the electrons broken bond are then used to form a new carbon-carbon double bond (C C) Part B 2-Bromo-2-methylbutane undergoes an El elimination reaction in the presence of ethanol. In the next reaction only one of the possible products is represented. Although the product shown is not the major product of the reaction, notice that there is more than one way t can be produced. Interactive 30 display mode Draw the missing intermediate and complets the mecharism (using eledtron-flow arrowa) in the second box. (You can automatically show hydrogen atoms by dicking the He buton, but you only need to explicitly show hydrogen atoms that participate in the mechanism) Orasw all missing reactants andlor products in the appropriate boxes by placing atoms on the grid and where needed on an stom, bond, or where cate the mechanism by drawing the electron-flow arrows on the molecules. Arrows should start on an atom or a bond and s conrecting them with bonds, including charges Indic a new bond should be created