PCL201H1 Lecture 11: Lecture 11 Clinical Pharmacology
Document Summary
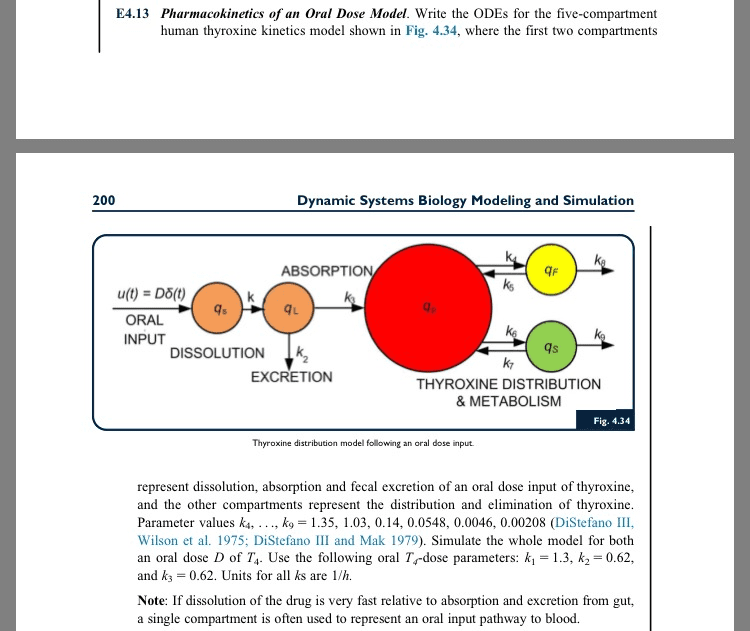
Bioavailability (f: fraction of uncharged drug reaching systemic circulation, measure as auc, measures extent of absorption and first pass, f = f(1-e) (f = absorption) [dr]plasma slow to increase (while absorption takes place) [dr]plasma decreases slightly as it equilibrates with all compartments. [dr]plasma rising slope not as steep as iv: capsule disintegrates, dissolves and absorbed via git. 1: equivalent iv/po dose po has lower cmax, po longer to reach cther after reaching cmax. Auc: []/time: extent of absorption, amount of drug in plasma bioavailability. Used to establish bioequivalence of generic drug: (auc trade)/(auc generic) must be 80- [drug] that reaches systemic unchanged: compare bioequivalence, measure extent of absorption compare routes of administration. Half life t time to decrease [drug] to half of previous value t = 0. 693/k. Rule of thumb: 5 half lives for 96. 9% drug removal. Reaching therapeutic window (tw) getting right [dr]plasma: cther to ctox: upper/lower boundaries of tw, css half-way between peak an trough.