Biochemistry 3380G Lecture Notes - Activation Energy, Enzyme, Enzyme Kinetics
Document Summary
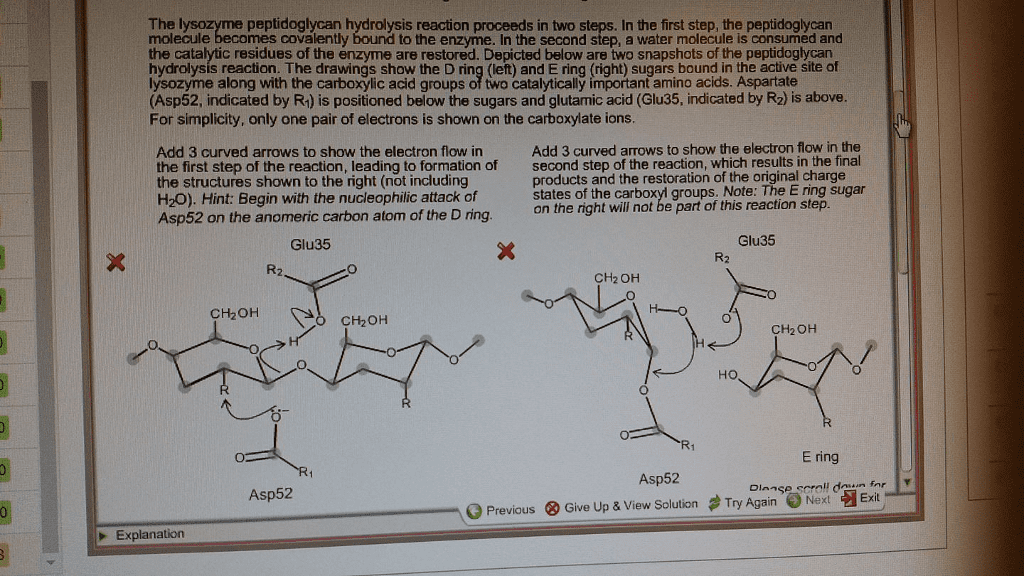
A small, protective enzyme found in tears, saliva, mucus, and eggs, lysozymes hydrolyze polysaccharides found in cell walls of some bacteria, which essentially breaks down their bacterial wall. They perform cleavage on the sugars of the cell wall. A part of the innate immune system, it is stabilized by disulphide bonds. The structure of the lysozyme contains two acidic residues (which are critical in allowing the enzymatic reaction to occur) and a small cleft for an active site. A chain of five sugar molecules (a, b, c, d, and e) is the substrate, which the lysozyme will act upon, forming the enzyme substrate complex (es complex). As this substrate approaches the lysozyme, it will often cause its active site or cleft to change slightly. In between sugars d and e, the lysozyme forces sugar d into a strained chair conformation. This makes it amendable for the next step of the reaction to occur.