Chemistry 1027A/B Lecture Notes - Stereochemistry, Vinyl Chloride, Atomic Number
Document Summary
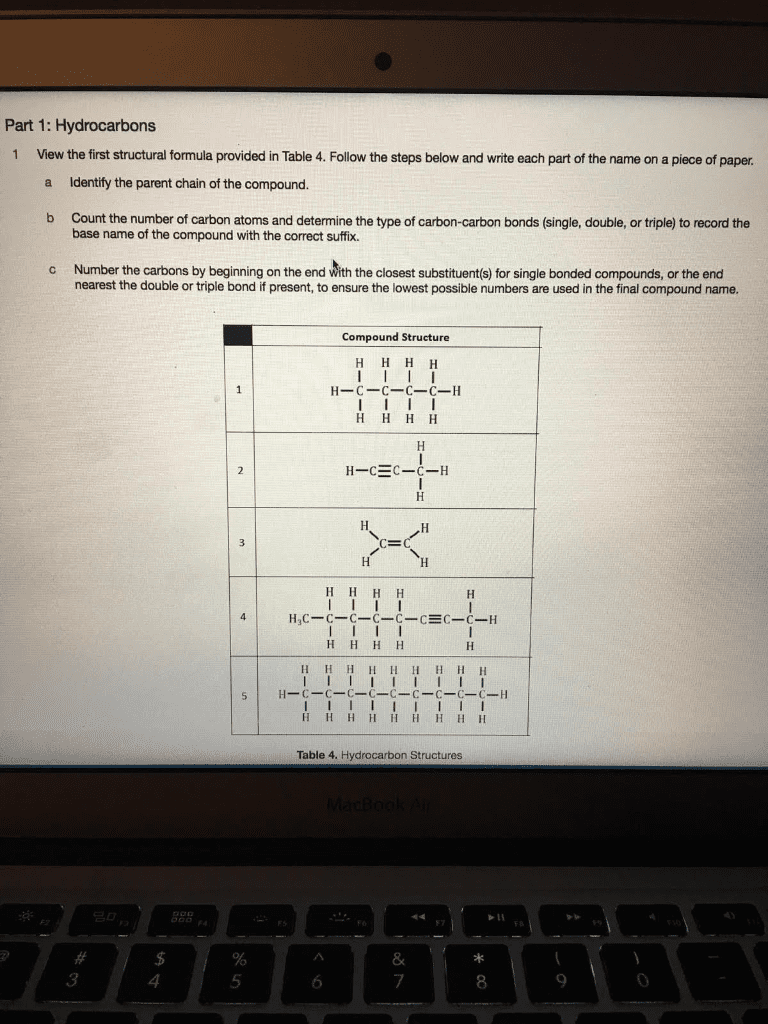
Organic chemistry key concepts: alkenes & alkynes (usselman version) The alkenes are hydrocarbons of the general formula cnh2n ; 2 less hs because of the presence of a double-bond. The alkynes have the general formula cnh2n 2 and contain a triple bond. Each reduction of 2 hydrogens in the general alkane formula represents one unit of unsaturation. Examples of one unit of unsaturation include a bond or a ring. Examples of 2 units of unsaturation include a bond in a ring or 2 bonds (in a triple bond). Isolated arrangements include 2 or more bonds between the bonds. Conjugated arrangements include multiple bonds which alternate with single bonds. Cumulated arrangements include two double bonds on a single carbon. For c = c, change the suffix to ene. For c = c, change the suffix to yne. The longest carbon chain must contain as many bonds as possible.