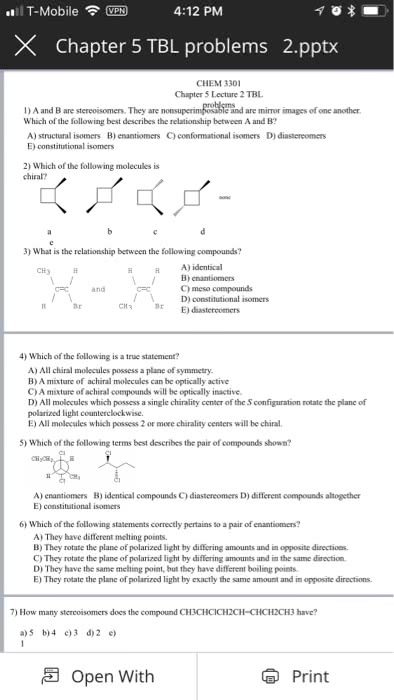
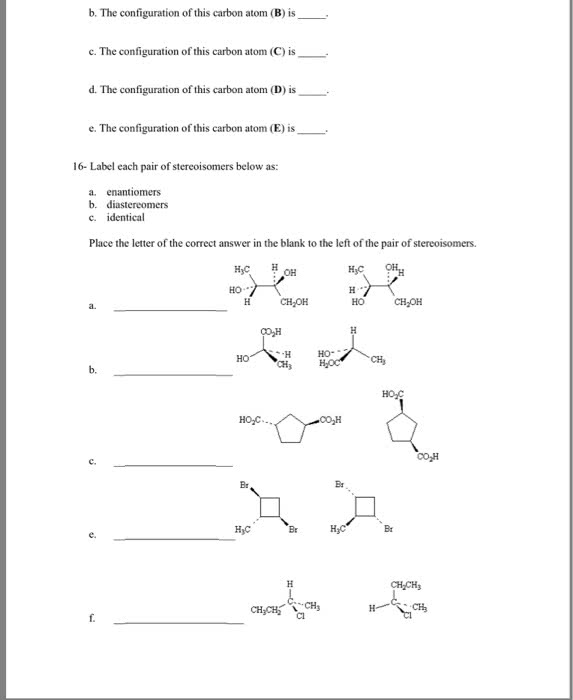
Chemistry 1027A/B Lecture Notes - Specific Rotation, Absolute Rotation, Stereocenter
Document Summary
A chiral object is any natural object which has no plane of symmetry. An achiral object is one with a plane of symmetry. All chiral objects are not superimposable on their mirror images. Conversely, all achiral objects are superimposable on their mirror images. The most common source of chirality in a molecule is a stereocentre, usually an atom such as carbon bonded to 4 different groups or substituents. All achiral molecules contain a plane of symmetry (generally true). The two mirror image forms of a chiral molecule are stereoisomers known as enantiomers. Enantiomers (stereoisomers) are different compounds (they do not interconvert at room temperatures) and their chemistry is exactly the same. They only differ in how they affect plane- polarized light and how they react with other chiral compounds. Stereocentres are commonly indicated with a * in a 3d molecular drawing. The r, s convention is utilized through the application of the same rules used for e, z isomers.