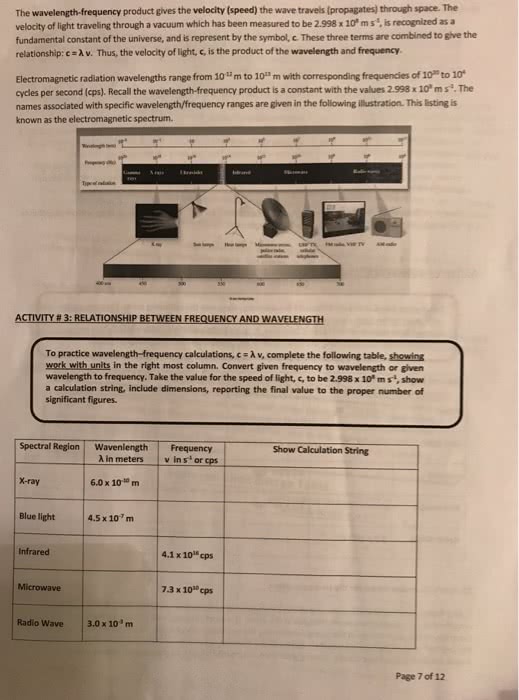
ZA Indicate the answer choice that best completes the statement or answers the question. 1. Erwin Schrödinger developed a model for the behavior of electrons in atoms that is known as quantum mechanics. Which of the following statements concerning this model isare CORRECT 1. The energy of an electron is quant 2. The energy of an 3. Electrons travel in circular orbits around a nucleus. tized. electron is equal to its mass multiplied by the square of its velocity a. I only b. 2 only c. 3 only d. 1 and 2 e 1,2, and 3 (mass 1673 x 10-27 kg) moving 2. For a proton (mass-i with a velocity of 2.83 x 1o1 m/s, what is the de Broglie wavelength of 2.8 (in pm)? a. 0.356 pm b. 3.56 pm c. 14.0 pm d. 7.15 pm e. 28.5 pm he-_ of a photon of light isä¸ä¸proportional to its fi a. energy, directly, inversely b. energy, inversely, directly c. velocity, directly, inversely d. intensity, inversely, directly e. amplitude, directly, inversely 3. T frequency and proportional to its wavelength. 4. How many orbitals have the following set of quantum numbers n-2,-1, m.-17 5. Which of the following regions of the electromagnetic spectrum has the lowest frequency? a. x-ray b. gamma ray c. ultraviolet d. infrared e. visible 6. Which of the following statements is/are CORRECT? FM radio waves have longer wavelengths than visible radiation and AM radio waves have shorter wavelengths than visible radiation. Magnetic resonance imaging (MRI) uses a mixture of x-rays and gamma rays. Exposure to ultraviolet (UV) radiation can lead to sunburns. 2. 3. c. 3 only d. I and 2 e. 1,2, and 3 a. I only b. 2 only Page 1