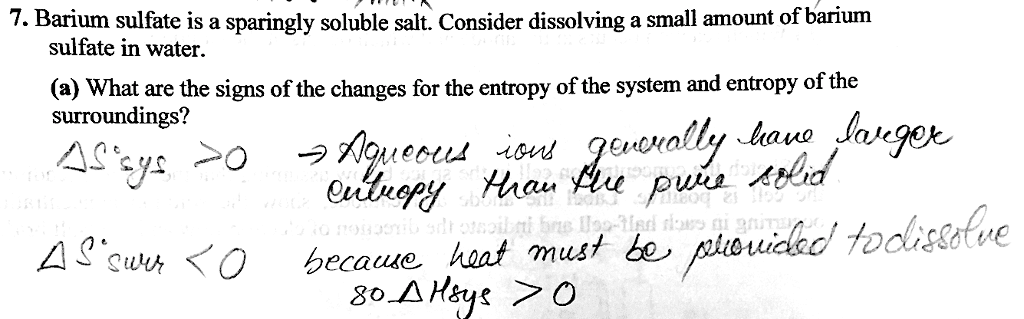Chemistry 1302A/B Lecture Notes - Lecture 6: Spontaneous Process, Randomness, Reversible Reaction
Document Summary
Free energy your system and your surroundings together must increase the entropy of the universe. However, we are usually interested in the system itself, not the universe. Free energy examines the spontaneity of the system specifically and does so in a way that automatically takes in the energy change of the surroundings. Hsystem ideally, this is negative and the reaction gives off heat (if you have a reaction that breaks a lot of bonds you have to put in energy therefore in order to achieve and exothermic, bonds are breaking) Ssys ideally, this is positive and the order is random thus, lets try to relate these two conditions in a way that will be able to predict spontaneity. Combining the equations for a spontaneous process ( s equations and h equations) we get. Hsystem - t ssystem <0 for a spontaneous process. H - t s <0 this must hold true.