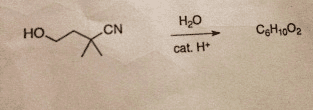Acid-catalyzed hydrolysis of the starting material below yieldsa product with the chemical formula C6H10O2. This product has astrong absorption in its IR spectrum at 1770 cm-1 and the followingsignals in its 1H NMR spectrum: 1.27 (singlet, 6H); 2.12(triplet,2H) and 4.26 (triplet, 2H). Determine the structure of theproduct and draw complete mechanism to show how it isformed.
This is to be around 12 steps apparently. All I need help withare the steps. Full points for thorough answer.
Here is the reaction:
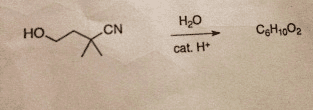
This is what the product should be:
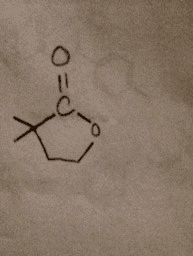
Acid-catalyzed hydrolysis of the starting material below yields a product with the chemical formula C6H10O2. This product has a strong absorption in its IR spectrum at 1770 cm-1 and the following signals in its 1H NMR spectrum: 1.27 (singlet, 6H); 2.12 (triplet,2H) and 4.26 (triplet, 2H). Determine the structure of the product and draw complete mechanism to show how it is formed. This is to be around 12 steps apparently. All I need help with are the steps. Full points for thorough answer. Here is the reaction: This is what the product should be: