CHM 113 Lecture Notes - Solvation, By2, Vaporization
Document Summary
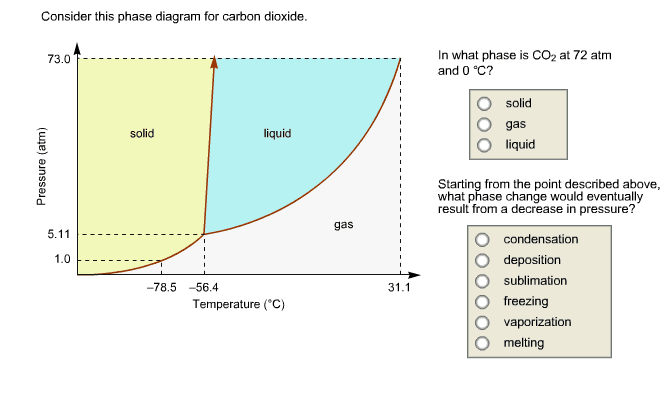
Critical temperature: the highest temperature at which a distinct liquid phase can form. Critical pressure: the pressure required to liquefy a gas at critical temperature. Phase diagram: temperature/pressure plot that summarizes the conditions under which different phases of matter occur. Critical point (b): critical temperature and pressure. Triple point (a): all three phases of matter all present in equilibrium with one another. Solution: homogenous mixture of two or more substances. Solvent: substance present in greatest amount: if the solvent is water, the solution is aqueous. Ion-dipole forces: if the solvent is h2o, this step is called hydration.