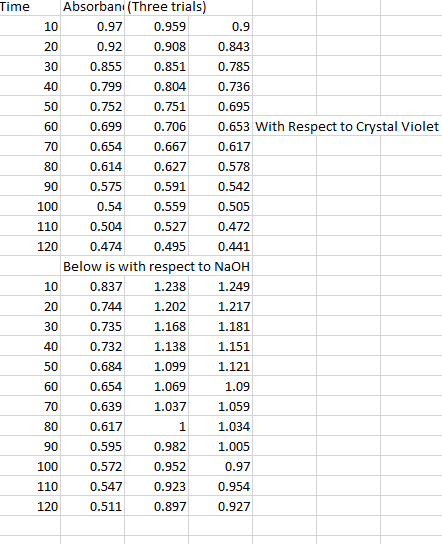CHEM 1111 Lecture Notes - Lecture 14: Potassium Dichromate, Reaction Rate, Reagent
41 views2 pages
Document Summary
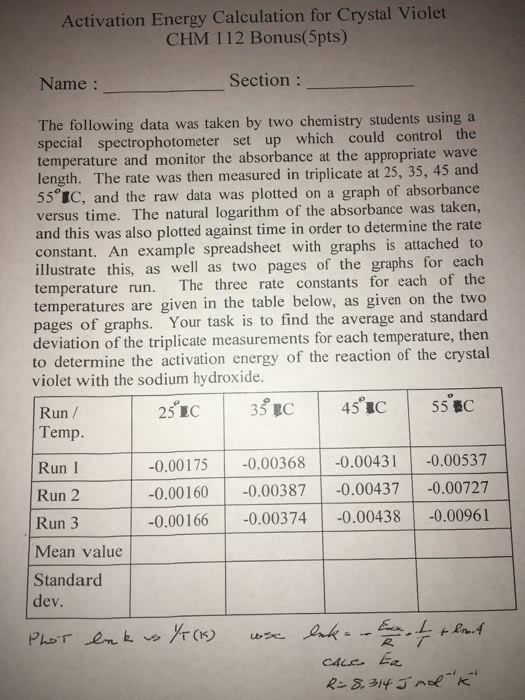
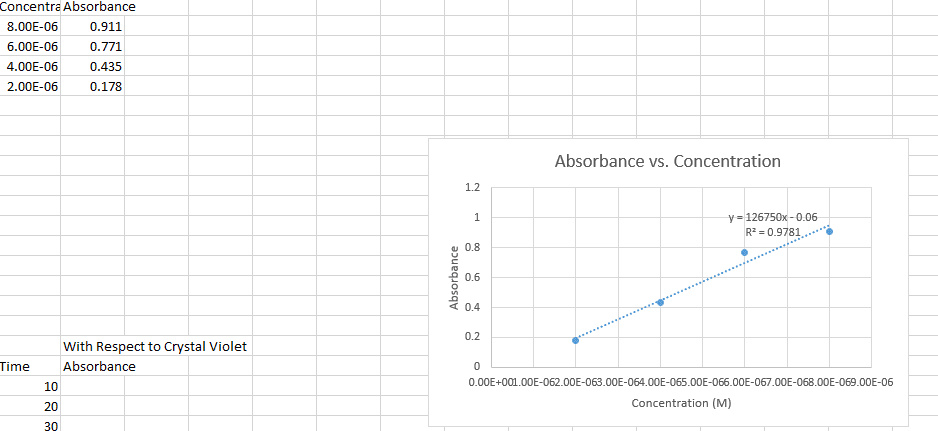
Calculations: see attached sheets, the data shows that the reaction is first order with respect to the dichromate. The units are s-1 because in a first order reaction rate (m/s) is equal to k times the concentration of the reactants (m). Therefore k must have units of s-1: rate constant: (1 / m120) = kt + (1 / m0) for t = 120s so, k = ((1 / m0) (1 / m120)) / 120. Questions: the rate of the reaction would be altered if the initial concentration of potassium dichromate in solution were changed. This could be done by changing the amount of 0. 0196 m potassium dichromate which is added to the erlenmeyer flask. Incorrectly measuring the amounts of solute and solvent could affect the experiment because not enough of the reagent would cause the reaction to not reach completion.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232