CHEM 101 Lecture Notes - Lecture 11: Phosphorus Pentoxide, Molar Mass, Chemical Formula
Document Summary
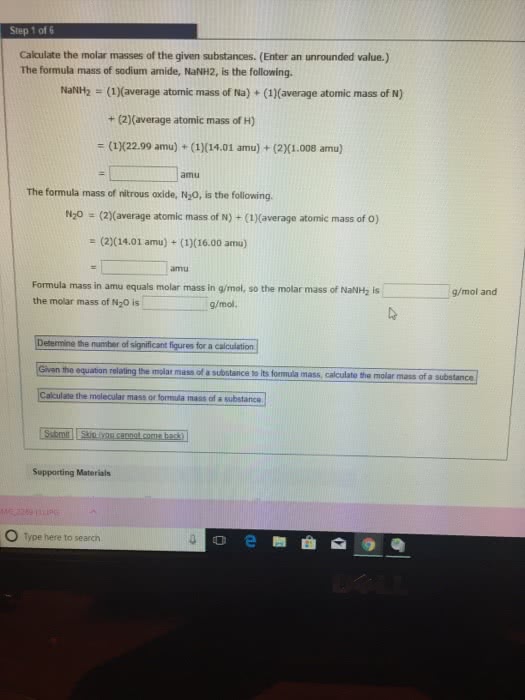
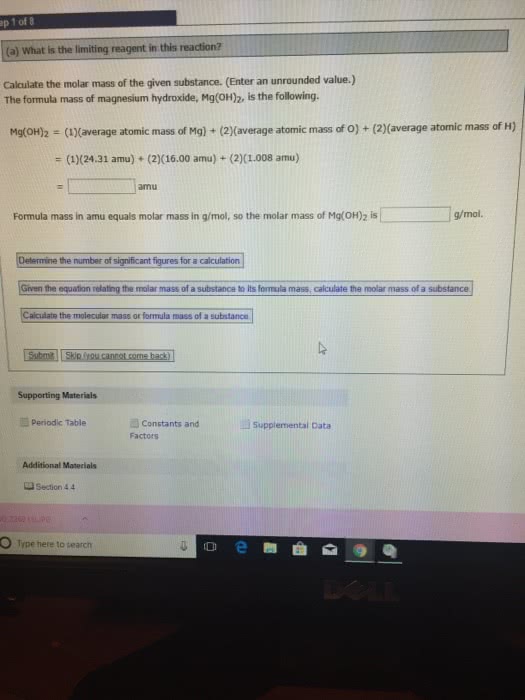
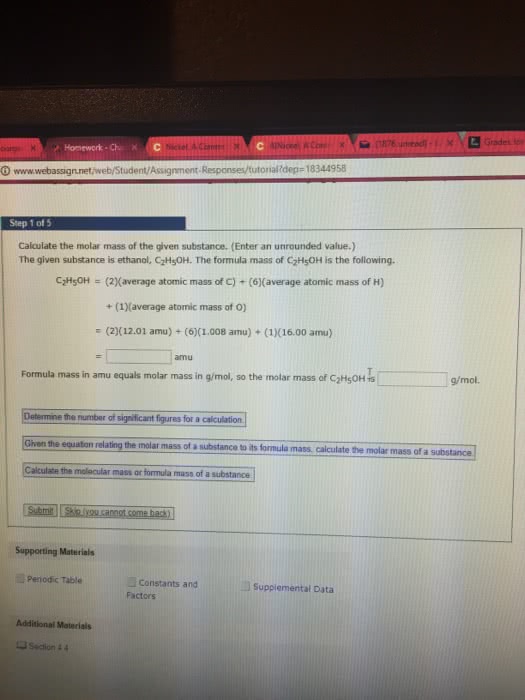
Review from chapter 3: molecular mass: the sum of the atomic masses of all the atoms represented in the chemical formula of a substance. The mole: the chemist"s counting unit is the mole(mol), which represents the number 6. 002x10^23, this is called avogadro"s number. It can be used as a conversion factor between moles and numbers of particles (1 mole c = 6. 022x10^23 atoms carbon: the molar mass of a compound has the same numerical value as the molecular mass, except the units are g/mol rather than amu. Molar mass: mass in grams of one mole of the substance: Molar mass of n = 14. 01 g/mol. Chemical formula - microscopic view: the numerical subscripts in a chemical formula give the number of atoms of the various elements present in 1 formula unit of the substance. In one molecule of p2o5, two atoms of phosphorus and five atoms of oxygen are present.