CHEM 116 Lecture Notes - Lecture 19: Gibbs Free Energy, Chemical Equation, Spontaneous Process
Document Summary
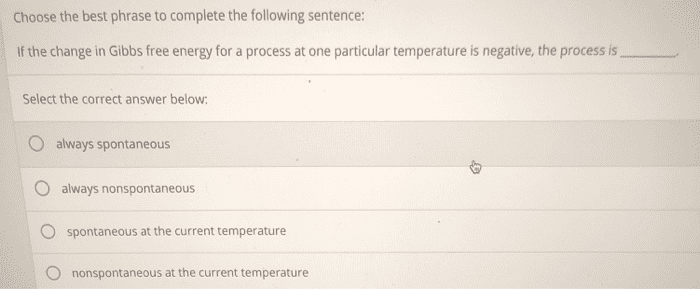
The reaction is nonspontaneous at low temperatures but spontaneous at high temperatures. 19. 7: free energy changes in chemical reactions: calculating grxn. Ways to calculate the standard free energy changes: g rxn = h rxn -t s rxn, g rxn = (delta g products)- (delta g reactants) A process that is nonspontaneous can be made spontaneous by coupling it with another process that highly spontaneous. 19. 8: free energy changes for nonstandard states: the relationship between dg rxn and dgrxn. We can calculate the free energy change of a reaction under nonstandard conditions (dgrxn) from this equation: dgrxn= dg rxn + rt lnq, q is the reactant quotient. 19. 9: free energy and equilibrium: relating dg rxn to the equilibrium constant (k) When k<1, ln k is negative and dg rxn is positive. Q=1) the reaction is spontaneous in the reverse direction. When k>1, ln k is positive and dg rxn is negative. Q=1) the reaction is spontaneous in the forward direction.