CAS CH 102 Lecture Notes - Lecture 15: Partial Pressure, Joule
Document Summary
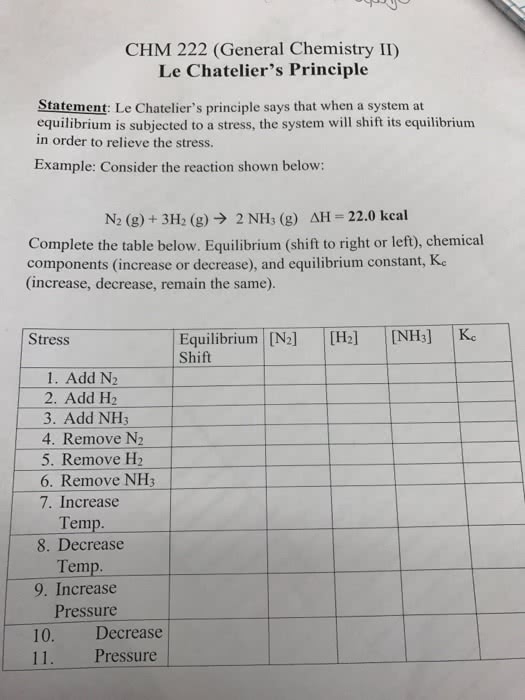
Chemical equilibrium has been reached in a reaction when the rate of the forward reaction is equal to the rate of the reverse reaction. When a chemical reaction has reached equilibrium, collisions are still occurring: the reaction is now happening in each direction at the same rate. This means that reactants are being formed at the same rate as products are being formed, and this is indicated by double arrows, At equilibrium, the reaction can lie far to the right, meaning that there are more products in existence at equilibrium, or far to the left, meaning that at equilibrium there are more reactants. Le chatelier"s principle states that when a system in chemical equilibrium is disturbed by a change of temperature, pressure, or a concentration, the system shifts in equilibrium composition in a way that tends to counteract this change of variable.