CAS CH 171 Lecture 2: Pre-Lab December 5, 2016
Document Summary
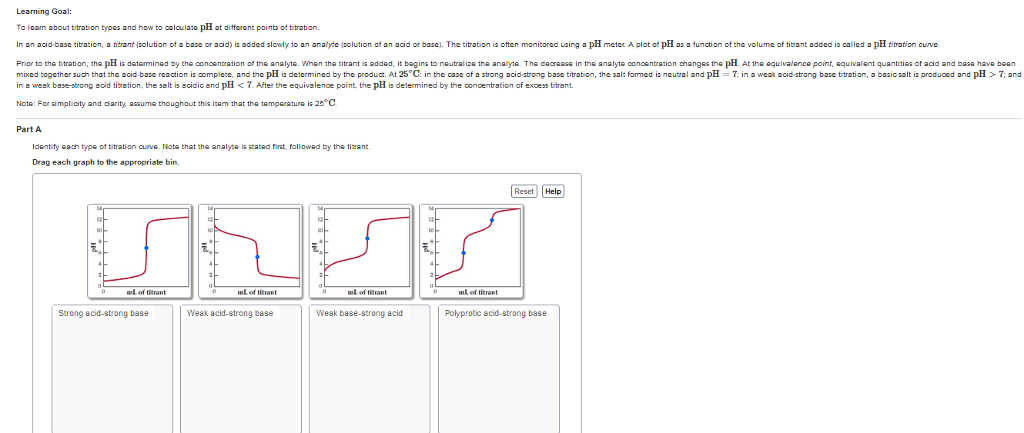
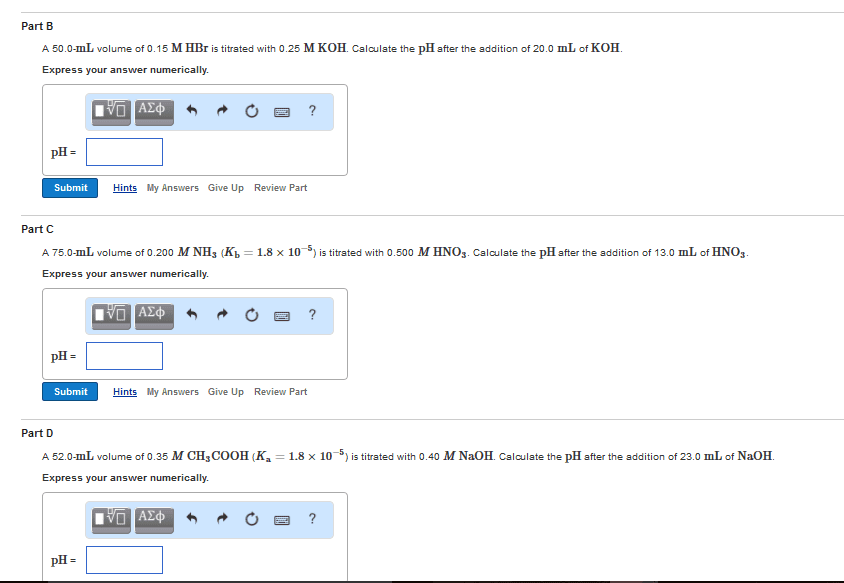
Titration- measurement of the volume of a standard solution required to completely react with a measured volume or mass of substance being analyzed (a. k. a. analyte) Standard solution- solution containing a known concentration of solute. Standardization- process of determining the concentration of a solution by using it to titrate a carefully measured quantity of a reference standard. Indicator- a substance added to an analyte solution in order to give an observable physical change (end point) at or near the equivalence point. Equivalence - the point at which an added titrant is stoichiometrically equal to the number of moles of analyte present in the sample. Normality- another way of expressing the concentration of a solution; it uses the alternative chemical unit of mass called equivalent weight. A titration curve provides a visual picture of how a property of the titration reaction changes as we add titrant to the analyte. All of a sudden see a big change.