CHEM 113 Lecture Notes - Equilibrium Constant, Reaction Rate Constant, Stoichiometry
Document Summary
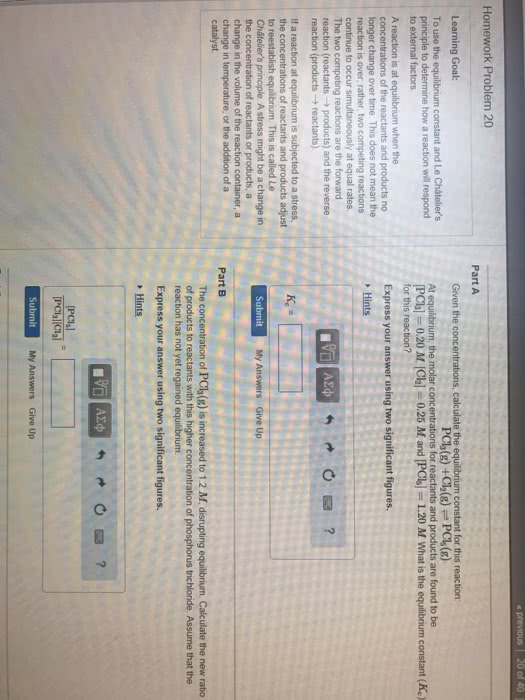
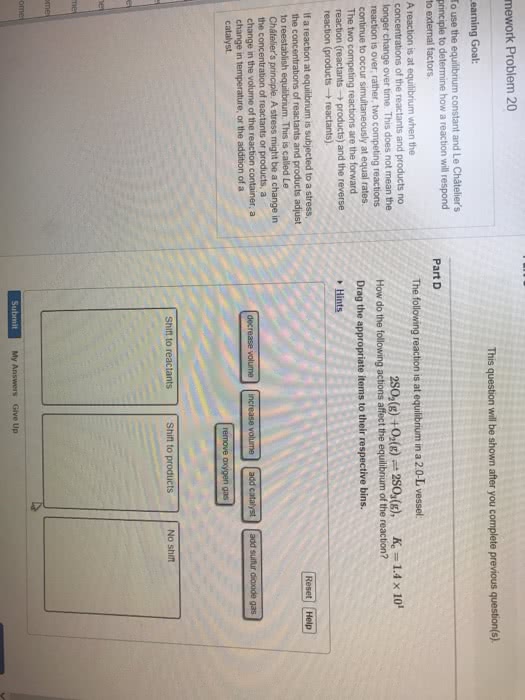
After a sufficient amount of time, a chemical system will approach a point where the concentration of reactants and products no longer change equilibrium. Equilibrium constant can be written straight from stoichiometry. K is a ratio of equilibrium concentrations (product to reactant) at a particular temperature. It is the opposing rates that are equal at equilibrium, not necessarily the concentrations. The magnitude of k is an indication of how far a reaction proceeds to product at a given temperature. Compounds a, b, and c react according to the following equation. 3a (g) + 2b (g) 2c (g) At 100 c a mixture of these gases at equilibrium showed that [a] = 0. 855 m, [b] = 1. 23 m, and. For an overall reaction, find k for individual steps and multiply them together. If you take the same reaction and multiply the whole thing by 2, the k is raised to the second power.