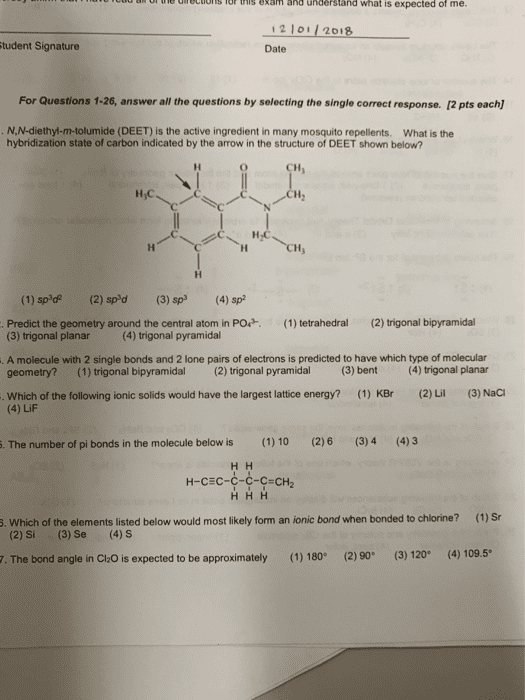
e ecios or this exam and understand what is expected of me. 12 1011 2018 tudent Signature Date For Questions 1-26, answer all the questions by selecting the single correct response. [2 pts each) N.N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents. What is the hybridization state of carbon indicated by the arrow in the structure of DEET shown below? CH H,C CH (1) sp'd(2) spd(3)p(4) sp Predict the geometry around the central atom in PO. (1) tetrahedral (2) trigonal bipyramidal (3) trigonal planar(4) trigonal pyramidal A molecule with 2 single bonds and 2 lone pairs of electrons is predicted to have which type of molecular geometry?(1) trigonal bipyramidal (2) trigonal pyramidal (3) bent (4) trigonal planar Which of the following ionic solids would have the largest lattice energy? (4) LiF (1) KBr (2)Li (3) NaC . The number of pi bonds in the molecule below is (1) 10 (2)6 (3)4 (4)3 ÐÐ Ð-СÐС-С-С-С-СÐÐ 5. Which of the elements listed below would most likely form an ionic bond when bonded to chlorine? (1) Sr (2) Si (3) Se (4)S 7. The bond angle in CizO is expected to be approximately(1)180 (2)90 (3) 120 (4) 109.5*
Show transcribed image text e ecios or this exam and understand what is expected of me. 12 1011 2018 tudent Signature Date For Questions 1-26, answer all the questions by selecting the single correct response. [2 pts each) N.N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents. What is the hybridization state of carbon indicated by the arrow in the structure of DEET shown below? CH H,C CH (1) sp'd(2) spd(3)p(4) sp Predict the geometry around the central atom in PO. (1) tetrahedral (2) trigonal bipyramidal (3) trigonal planar(4) trigonal pyramidal A molecule with 2 single bonds and 2 lone pairs of electrons is predicted to have which type of molecular geometry?(1) trigonal bipyramidal (2) trigonal pyramidal (3) bent (4) trigonal planar Which of the following ionic solids would have the largest lattice energy? (4) LiF (1) KBr (2)Li (3) NaC . The number of pi bonds in the molecule below is (1) 10 (2)6 (3)4 (4)3 ÐÐ Ð-СÐС-С-С-С-СÐÐ 5. Which of the elements listed below would most likely form an ionic bond when bonded to chlorine? (1) Sr (2) Si (3) Se (4)S 7. The bond angle in CizO is expected to be approximately(1)180 (2)90 (3) 120 (4) 109.5*