CHEM 120A Lecture Notes - Lecture 10: Thermochemistry, Kinetic Energy, Potential Energy
Document Summary
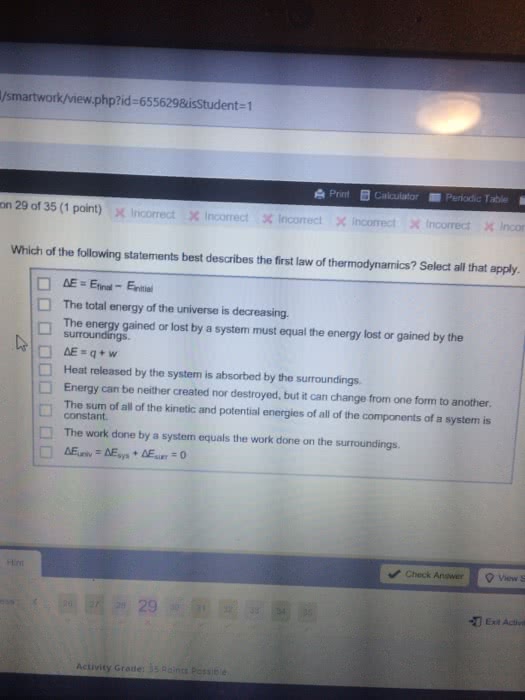
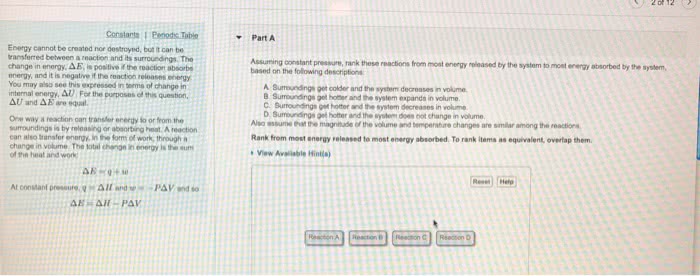
According to the law of conservation of energy, energy can neither be created or destroyed in a reaction. System: a point of reference: in thermochemistry, reactions will be the system studied. Surroundings: the environment surrounding the system that can exchange energy: if something leaves the system, it goes to the surroundings, a system plus its surroundings is the universe. State functions do not depend on the path taken: real-life example: you need to climb a mountain but have se(cid:448)eral paths of doi(cid:374)g so. It does(cid:374)"t (cid:373)atter (cid:449)hich o(cid:374)e you take since they will all take you up: back to chemistry: internal energy is the sum of the kinetic and potential energy of the system. The change in internal energy of a system only constitutes the energy at the end of the reaction subtracted by the energy at the beginning of the reaction. It can also be thought of as the difference in energy between the products and reactants.