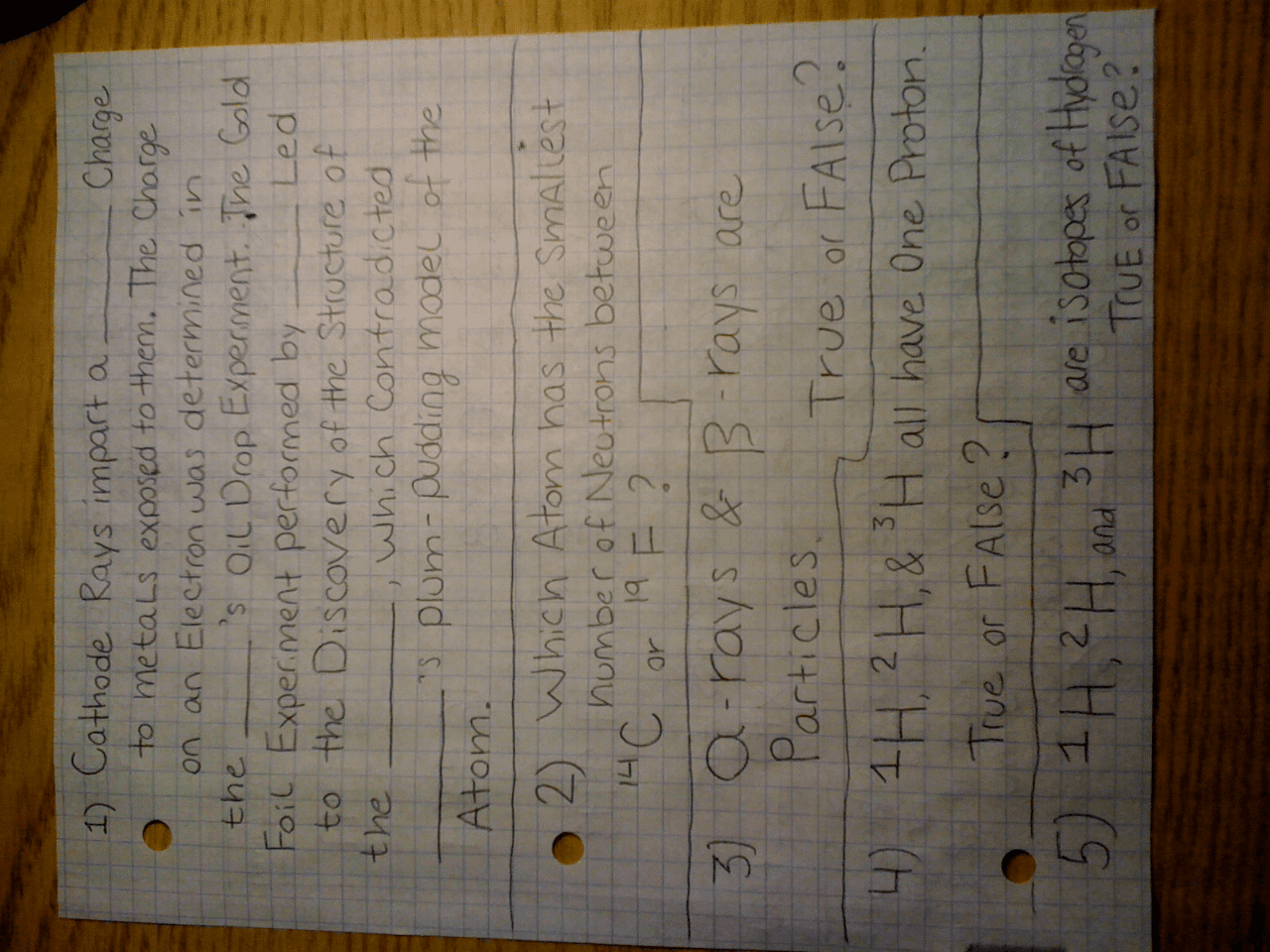CHEM 1021 Lecture Notes - Lecture 7: Cathode Ray Tube, Radiant Energy, Picometre
Document Summary
Thomson"s cathode ray tube experiment led to the plum pudding atomic model in 1897 (discovery of the electron) Rutherford"s gold foil experiment led to the nuclear atomic model in 1909 (nucleus as the positively charged center of the atom) Light seen with your eyes is visible light (a form of electromagnetic radiation, henceforth emr) Emr is energy moving through space as waves (thus, radiant energy) Light is one of many types of emr. Emr as energy propagated through space as waves by electric and magnetic fields. Characteristics of waves (the crest of a wave is the peak, the trough is the low point): Wavelength (the distance between successive peaks; measured by the length of 1 repeat; symbolized by lamda ) Frequency (the number of complete wavelengths that pass a point in 1 second; symbolized by nu ) Amplitude (the height of wave; symbolized by a) Emr as a continuum of wavelengths; long (10^3 m) to short (10^-12 m; picometer)