CHEM 101 Lecture Notes - Lecture 11: Ideal Gas Law, Ideal Gas, Gas Constant
57 views2 pages
4 Dec 2016
School
Department
Course
Professor
Document Summary
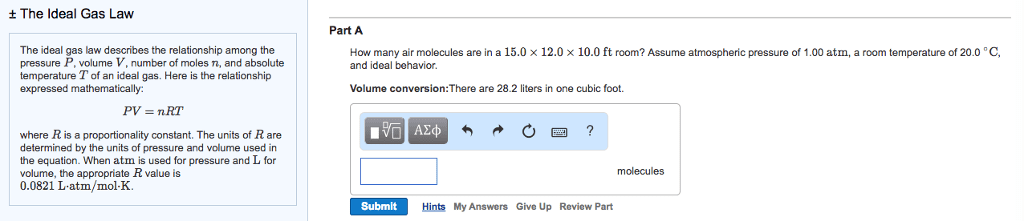
Exert a force when it collides with a surface or other gaseous particles. R is the gas constant and is the same for all gases. Used to define a volume of an ideal gas. Stp= standard temperature and pressure (not provided on the exam) Volume of 1mol of gas at stp. Volume of a gas at stp is called the molar volume. Notes: only need to know the temperature, molecular mass, moles and pressure to find (volume is unnecessary) Few gases are pure, most are mixture so the mixture can be thought of as one gas. Using percent by volumes of the gases in the mixture, one can find the partial. Dalton"s law pressures of the components in the mixture. The ratio of partial pressure a single gas contributes and total pressure is. The partial pressure of a component of the gas is its mole fraction multiplied by the total pressure.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232