Please help me with the results section! I posted the GC reports Incase you need them, but I also posted the results of the experiment. I just donât understand how to do the graph and the calculations they are asking for !
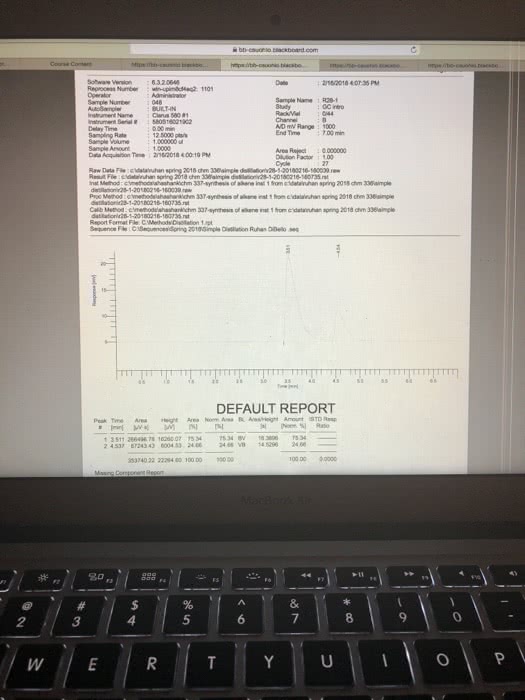
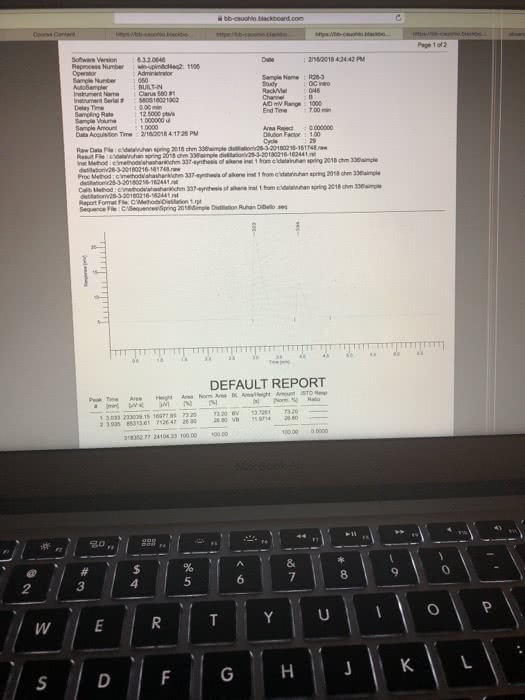
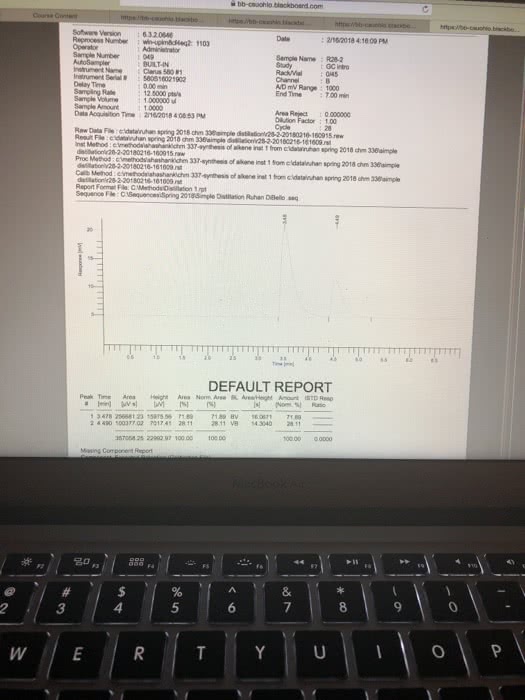
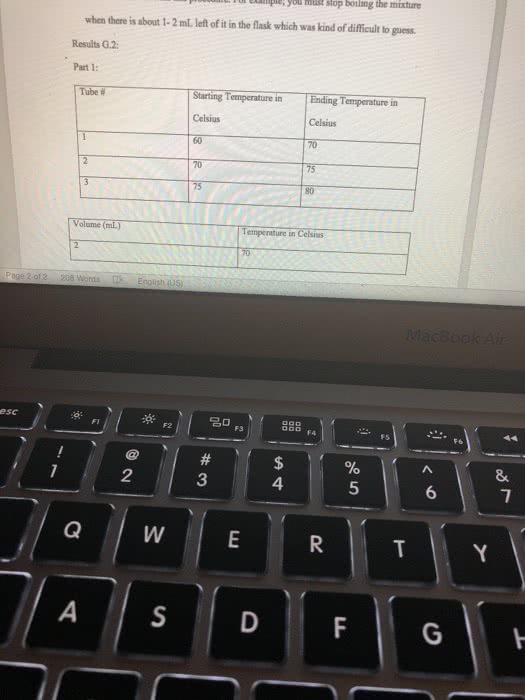
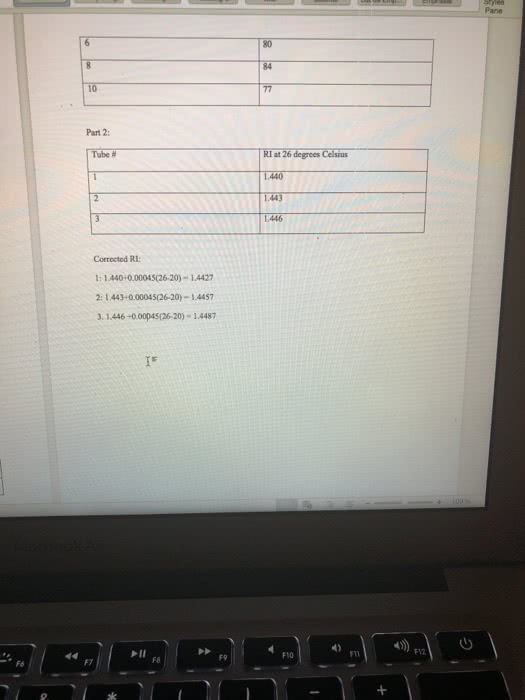
Results: Report all of your data acquired here, no data discussion Use the measurements recorded during your experiment they will be compared to the data you left behind or your TAs. Report your all data in a clear way Report the BP for pure Cyclohexane and pure Toluene. Use table for for your reporting your data: fraction number, BP range, RI (cnude and corrected); include table Build a graph using Excel or another software that allows for fit regression. In the total absence of toluene in the distilling mixture, your sample will be composed of 100% Cyclohexane. Therefore, the refractive index of the sample is equal to RI for pure Cyclohexane. In the total absence of CyclohexoneãCyclohexane), your sample is composed of toluene only and the refractive index is the one for pure toluene. Using the information above, create a groph of Refractive Index X Percentage of Cyclohexane. Use linear regression and identify the equation describing the line obtained. Groph, linear regression and equation must be presented on your report. Then Use the equation determined in the previous step to estimate the % of Toluene and Cyclohexane for each of the fractions collected in your distillation. Report your data in a table, include legend. For the Gas Chromatography experiments, use the to calculate the percentage of Cyclohexane and Toluene in each sample. Report your data in tables Show work for all the calculations and do not forget measurement units. Be clear on which calculations you are showing and what your mumbers represent. For example, when calculating the density of an organic liquid describe density mass organic liquid wolume of organic liquid, then proceed to your calculations. Any chemical structure must be done using ChemDraw or it will not be gtyn credit. (10/30 points) TAs: give 4 polnts for clearly reported calculations, with units, including correction of RI, percentage of solvents in both distillation and percentage of solvents from GC; 3 points for data presentation and 3 points for RI graph. Data shall match the data left behind or no credit will be given; Discussion: Disecussion is dissernarive. Use "le insead of SheiHe when writing the discnossion Your discussion shail expliain fhot necessardly in the seqaence belaw . What is distiliation? What is the difference btween simple and fracrional disnllation? e Using your lonwdedge of partial apor pressune and the pure roes boidling points, eküch solven What is the reiationshp of volality Boaling Poust and vopor pressure? you expect so usrit first cclonerane and doese Compore the BP of pure solvents to the BP range for each fraction, does it indicate pure jracrion for sach for borky solvents were obuined? What is Refractive Index and why to se it for the anaiss of distillation pactions? Do your R results, including the calculation of % of eyciaherare and toluene, support pure fractions of each sohenz? of 2 821 Words English ( 4 esc æ¼ :