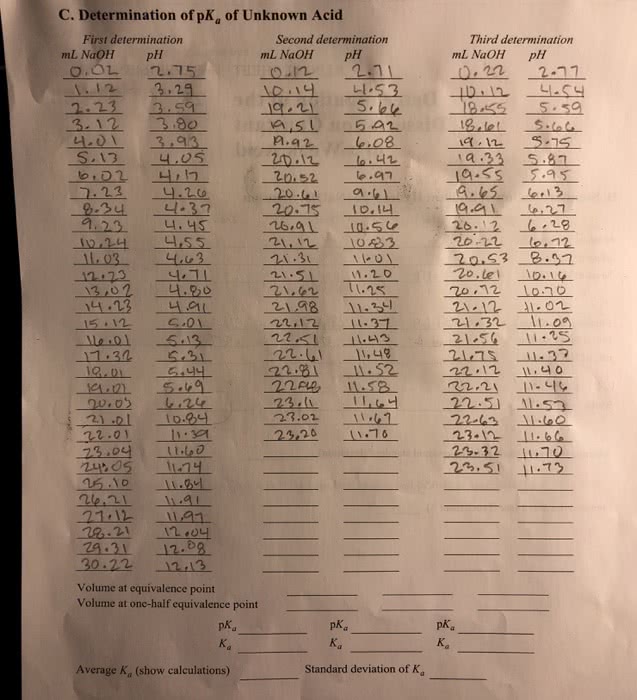
SEST-559 Lecture Notes - Lecture 4: Calcium Chloride, Water Softening, Burette

HARDNESS OF WATER REPORT SHEET
Name: ________________________________________ Date: _____________
Laboratory instructor or teaching assistant: _________________________________________
Part A: Preparation of Calcium Carbonate Standard
a) Mass of calcium carbonate ______________(g)
b) Total volume of standard solution ______________(L)
c) Molarity of calcium carbonate standard ______________(M)
Part B: Determination of Blank Volume
Trial 1 Trial 2 Trial 3
Final Burette Reading (mL)
Initial Burette Reading (mL)
Volume for Blank (mL)
Average Blank: ___________±___________
Part C: Standardization of EDTA Titration
Trial 1 Trial 2 Trial 3
Final Burette Reading (mL)
Initial Burette Reading (mL)
Volume for EDTA (mL)
Average Volume of Blank (mL)
Volume of EDTA used to titrate Ca2+
Molarity of EDTA (show calcs)
Average Molarity of EDTA (show calc): ___________±___________
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Part a: preparation of calcium carbonate standard: mass of calcium carbonate, total volume of standard solution, molarity of calcium carbonate standard. Part d: determination of water hardness in unknown sample. Note: show all calculations for trial #1 on a separate page. Volume of edta used to titrate blank (ml) Volume of edta required to titrate unknown sample (ml) Moles of edta required to titrate 25 ml of unknown sample. Moles of caco3 per 25 ml of unknown sample (moles) Weight of caco3 per 25 ml of unknown sample (g) Weight of caco3 per 106 ml unknown sample (g) ppm of caco3 in unknown. The average caco3 content of unknown # is _____ _____ ppm. Please type these answers on a separate page: please create a results section for this experiment utilizing proper tables that are computer generated to show the data that you collected. This results section should be created as if this were a formal report.