CHEM 1211K Lecture 2: chapter two Atoms and elements
Document Summary
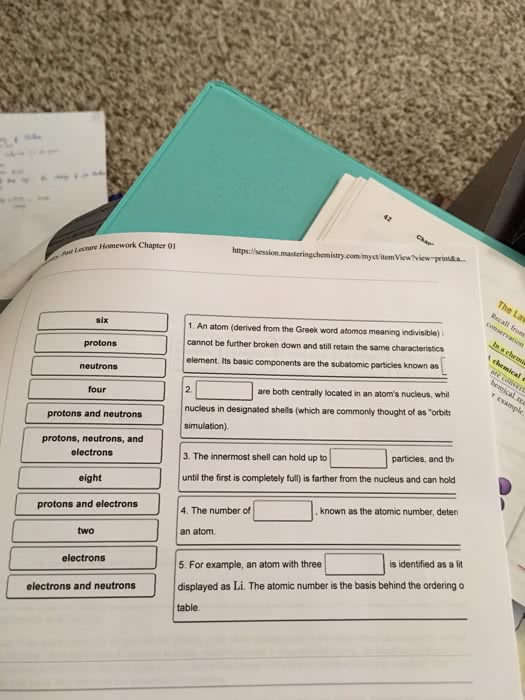
Check your current model: draw a carbon atom. There are three types of subatomic particles that will make up our atomic model: ____________, ____________, _____________ Protons and neutrons are compacted together in what we call the _______________ of an atom. Electrons are distributed in space around the nucleus: they are moving very fast in a volume surrounding the nucleus. There are a few fundamental properties of nature: examples: gravity, magnetism, and mass. Another fundamental property in nature is ________________ ______________. Particles may or may not have electrical charge. There are two types of electrical charge; we arbitrarily call one type ______________ and the other type. Every thing we discuss in this course ultimately occurs because of the interaction of these two types of charges. Oppositely charged particles will accelerate toward one another if not held apart. Like charged particles will accelerate away from one another if not held together.