CHEM 1201 Lecture 8: Limiting Reagents
122 views3 pages
Document Summary
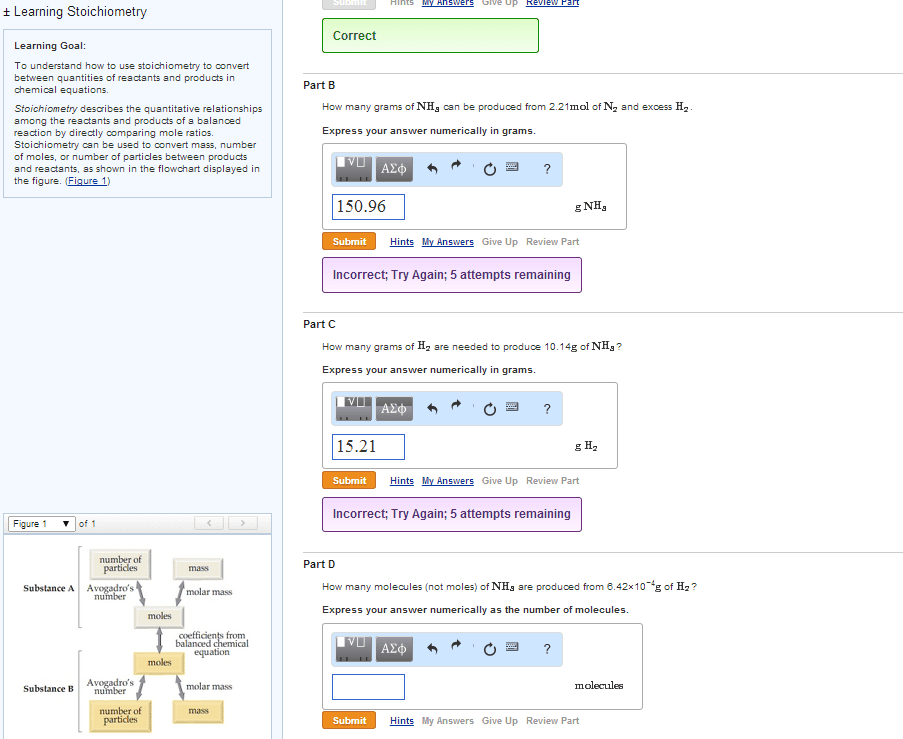
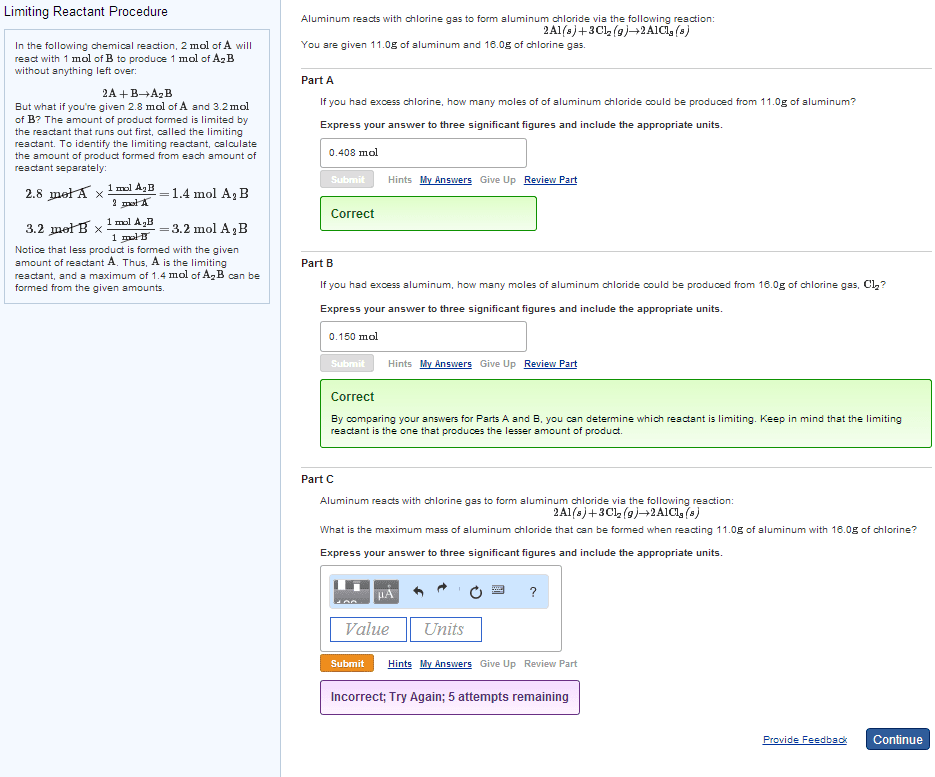
Chem 1201 - lecture 8 - limiting reagents. The amounts of two known reactants are given, one will be used up. The limiting reagent is completely consumed in the reaction. The limiting reagent limit"s the amount of product obtained. Therefore, at the end of reaction, those reactants present in excess will still be in the reaction mixture. Goal (#1): identify which reagent is the limiting reagent . Reagent amounts are given in gram amounts (haven"t covered molarity yet); so these values must be converted to moles via molar mass bearing in mind the stoichiometry of the reaction (most important!! The reagent with the smallest mole value, from using stoichiometry, is the limiting reagent. Multiply the moles of the limiting reagent times the molar mass of the product (and preserving stoichiometry) gives us the theoretical yield in grams. (i. e. moles x molar mass = grams).
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232