CHM 142 Lecture Notes - Ideal Gas Law, Reaction Quotient, Equilibrium Constant
Document Summary
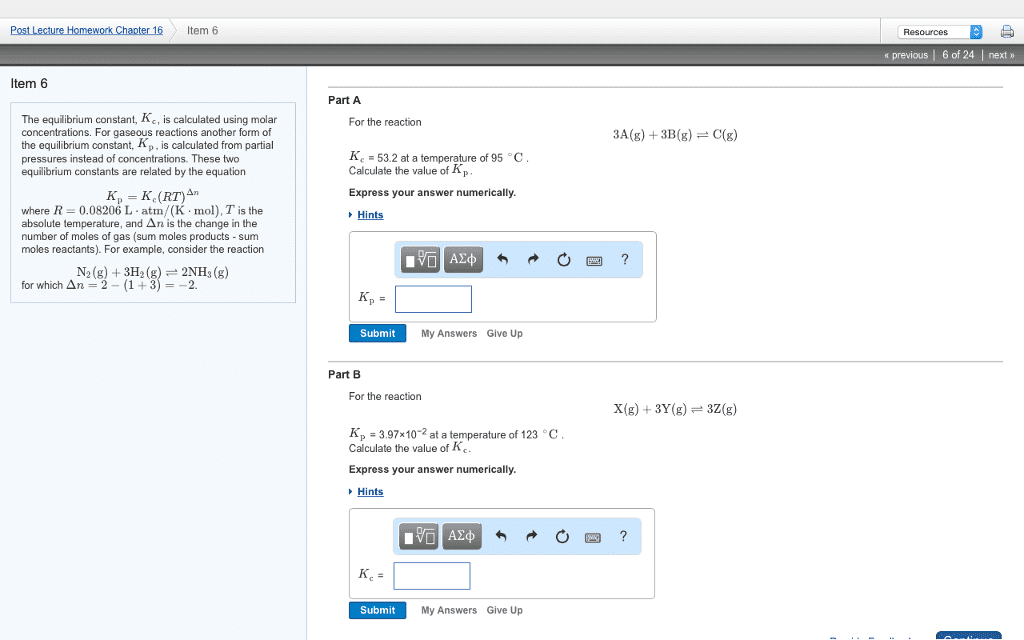
Chemical equilibrium a dynamic process in which concentrations of reactants and products remain constant over time. Concentrations are reactants and products are not necessarily equal at equilibrium. Rate of reaction forward is equal rate of reaction in reverse. Equilibrium constant (k) ratio of forward and reverse constants, or ratio of product to reactant concentrations (or partial pressures) for a reaction. K doesn"t tell you anything about the rate at which equilibrium is reached. Law of mass action equilibrium constant expression at chemical equilibrium has a characteristic value at a given temperature. P = (n/v)rt: n/v is molarity. Kp = kc(rt)^ n: n = number of moles of gaseous product moles of gaseous reactant, so, kp = kc when you have the same number of moles on both sides. K of a reverse reaction is the reciprocal of k for the forward reaction. K value for a reaction that is n multiple of the original reaction is original.