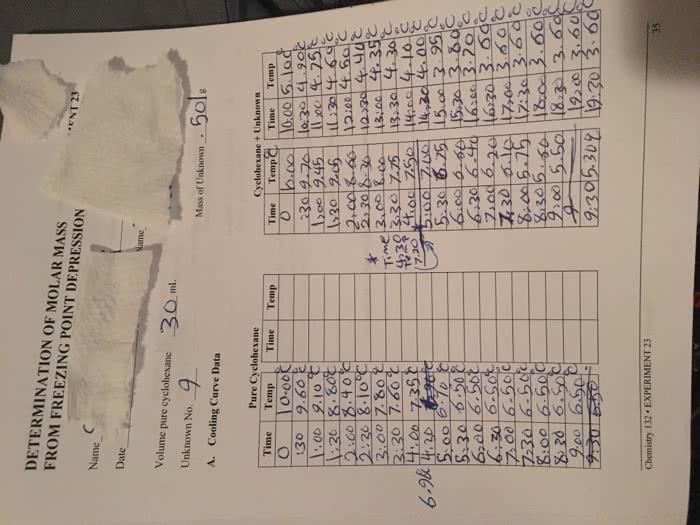
Experiment 10 Colligative Properties Introduction A pinch of wa make boiling water hotter. A handful will clear the snow from your steps. Every day we the physical properties of materials around us, but often don't know why we use these particular s or what chemistry is involved. Modifying the temperature of phase changes, by adding a second chemical to the pure solvent are examples of manipulating the Colligative Properties of a solution. this lab, we will study how varying the concentration and type of a salt affects the point of a solution. Colligative properties are properties of a solution containing 2 or more components. Typically, the primary is the solvent and the other called the such as color or hardness, properties depend on the number of particles of astute in the not the identity of the In many cases, the rokute will determine the number of solute particles (degree of ionization, solubility, etc,) in the solvent, but a colligative property does not depend upon the nature of the particles. Two colligative properties commonly encountered are boiling point elevation and freezing point depression (the temperature of of a is lower than of pure solvent). We will be studying boiling point elevation in this Most of the previous work that you have done with solutions has involved units of molarity, M, or moles of solute per liter of solution. Boiling point elevation calculations (as well as those for freezing point depression) or moles of solute per kilogram of solvent. boili change of temperature. When the temperature of a solution changes, the volume of the solution also changes. Since molarity depends on a change in wil the solution's molarity. Molality depends on the mass of the solvent. and thus is independent of temperature. n't Hoff Factor salt into and the of moles inorganic salts in an aque the based on the addition of the number dissociates, the total number of particles in the solution will be of cations and anions produced from the dissociation. Thus; Nacion Na Choaa (2moles of particles) Cacbrs Ca +2ch (3moles of particles) The quantity of particles produced per mole of solute is called the van'tHoff factor and is given the symbol, i, If 1 mole of sodium chloride were completely dissociated, iwould be 2. This theory works very well for dilute solutions and non-electrolytes (i 1, no dissociation at all since nonelectrolytes not ionize), For does not always apply to more concentrated solutions. Thus, when there are a large number of particles in a solution, they have a tendency to recombine as the salt and you don't get 100% dissociation. example, at a NaCl concentration of 0.1M, iwill be 1.81 (90% dissociation) rather than 2 (100%). Boiling Point Elevation solute causes the solution to boil at a higher When a solution is heated, the presence of a non-volatile point of the pure liquid and the temperature than the pure The difference between the boiling boiling point of the solution is: 169