 Help with these
Help with these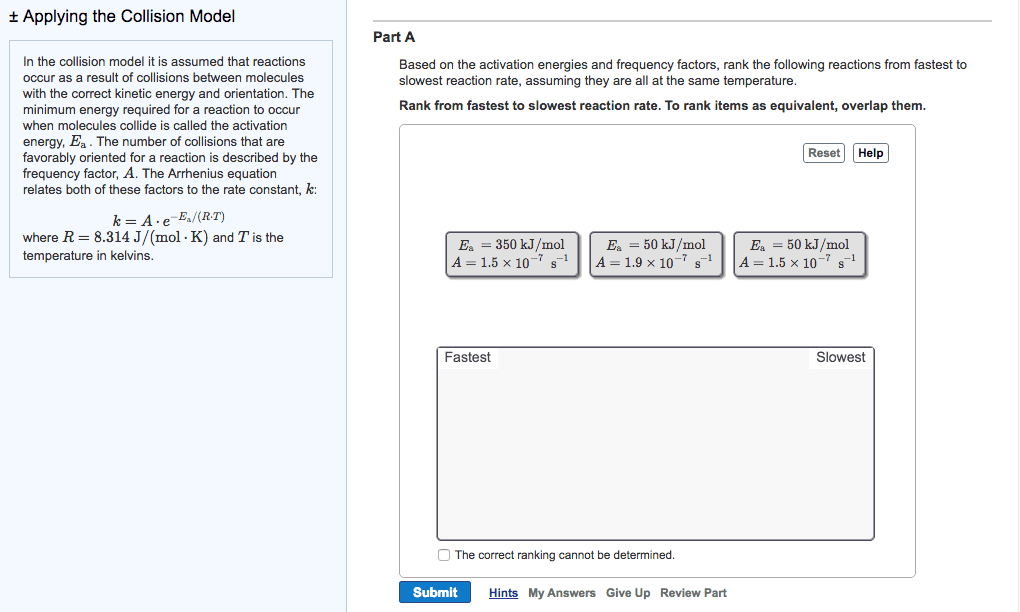
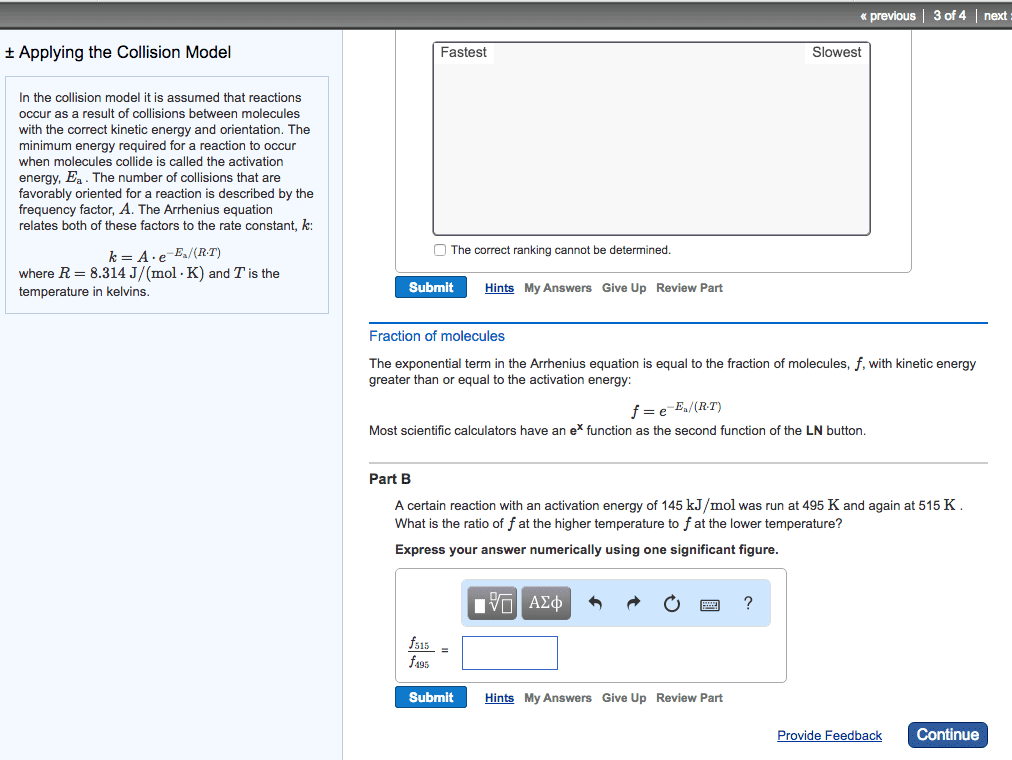
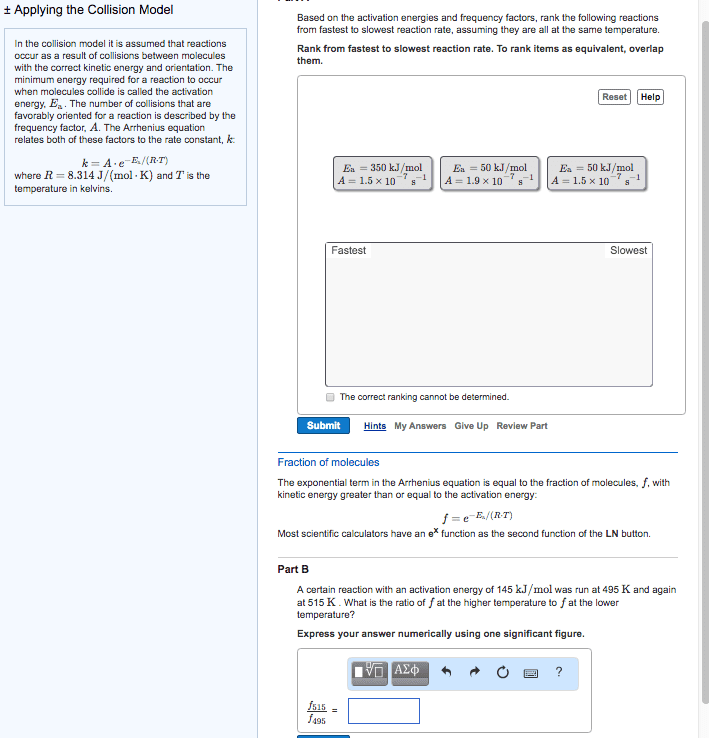
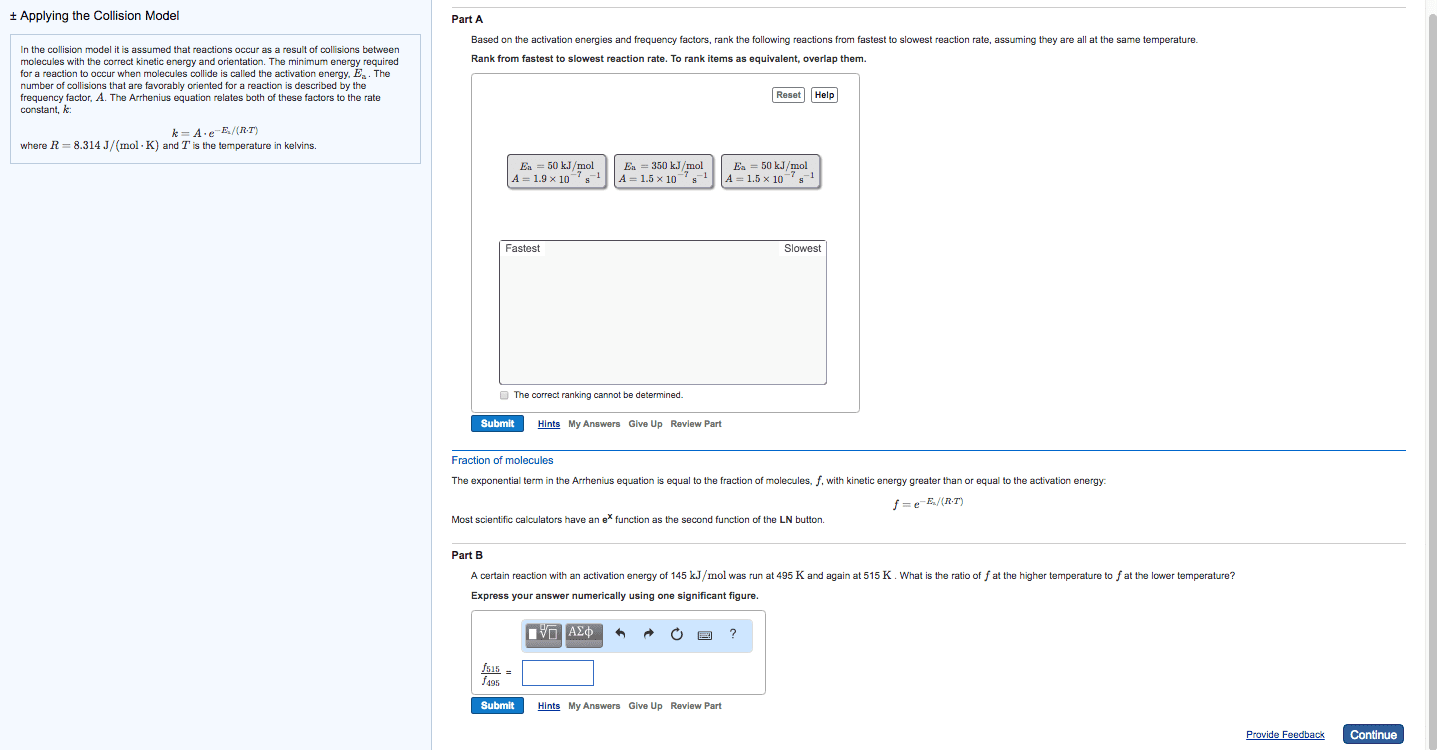
In the collision model it is assumed that reactions occur as a result of collisions between molecules with the correct kinetic energy and orientation. The minimum energy required for a reaction to occur when molecules collide is called the activation energy, E_a. The number of collisions that are favorably oriented for a reaction is described by the frequency factor, A. The Arrhenius equation relates both of these factors to the rate constant, k. k = A middot e^-E_a/(R middot T) where R = 8.314 J/(mol middot K) and T is the temperature in kelvins. Based on the activation energies and frequency factors, rank the following reactions from fastest to slowest reaction rate, assuming they are all at the same temperature. Rank from fastest to slowest reaction rate. To rank items as equivalent, overlap them. Fraction of molecules The exponential term in the Arrhenius equation is equal to the fraction of molecules, f, with kinetic energy greater than or equal to the activation energy: f = e^-E_a/(R middot T) Most scientific calculators have an e^x function as the second function of the LN button. Part B A certain reaction with an activation energy of 145 kJ/mol was run at 495 K and again at 515 K. What is the ratio of f at the higher temperature to f at the lower temperature? Express your answer numerically using one significant figure. In the collision model it is assumed that reactions occur as a result of collisions between molecules with the correct kinetic energy and orientation. The minimum energy required for a reaction to occur when molecules collide is called the activation energy, E_a. The number of collisions that are favorably oriented for a reaction is described by the frequency factor, A. The Arrhenius equation relates both of these factors to the rate constant, k: k = A middot e^-E_a/(R middot T) where R = 8.314 J/(mol middot K) and T is the temperature in kelvins. Part A Based on the activation energies and frequency factors, rank the following reactions from fastest to slowest reaction rate, assuming they are all at the same temperature. Rank from fastest to slowest reaction rate. To rank items as equivalent, overlap them. Fraction of molecules The exponential term in the Arrhenius equation is equal to the fraction of molecules, f, with kinetic energy greater than or equal to the activation energy: f = e^E_a/(R middot T) Most scientific calculators have an e^x function as the second function of the LN button. Part B A certain reaction with an activation energy of 145 kJ/mol was run at 495 K and again at 515 K. What is the ratio of f at the higher temperature to f at the lower temperature? Express your answer numerically using one significant figure. In the collision model it is assumed that reactions occur as a result of collisions between molecules with the correct kinetic energy and orientation. The minimum energy required for a reaction to occur when molecules collide is called the activation energy, Ea. The number of collisions that are favorably oriented for a reaction is described by the frequency factor, A. The Arrhenius equation relates both of these factors to the rate constant, k: k = A middot e^-E_a/(R middot T) where R = 8.314 J/(mol middot K) and T is the temperature in kelvins. Fraction of molecules The exponential term in the Arrhenius equation is equal to the fraction of molecules, f, with kinetic energy greater than or equal to the activation energy: f = e^-E_a/(R middot T) Most scientific calculators have an e^x function as the second function of the LN button. Part B A certain reaction with an activation energy of 14 kJ/mol was run at 495 K and again at 515 K. What is the ratio of f at the higher temperature to f at the lower temperature? Express your answer numerically using one significant figure.








