CHEM 1210 Lecture Notes - Lecture 5: Kinetic Theory Of Gases, Ideal Gas Law, Gas Constant
Document Summary
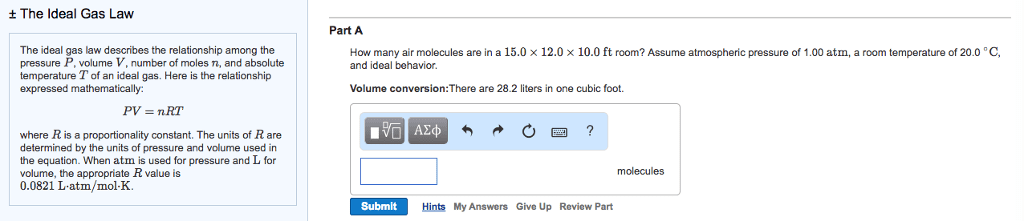
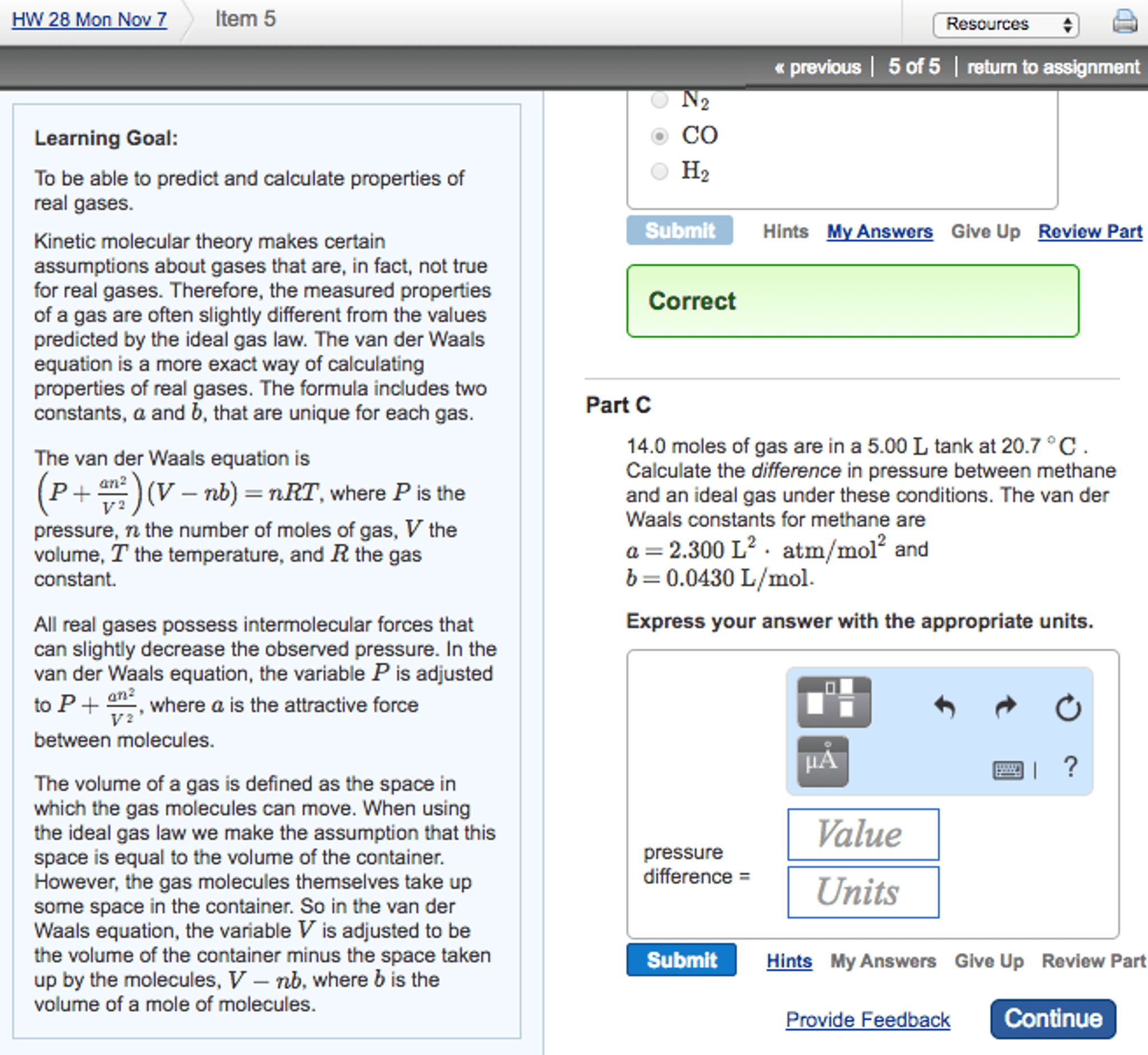
Avogadro"s law allows us to write a gas law that is valid not only for any. P, v, and t but also for any mass of gas. P = pressure of the gas in atmospheres (atm) V = volume of the gas in liters (l) n = moles of the gas (mol) or mass/molecular mass (m/m) R = ideal gas constant (r = 0. 0821 l-atm/mol-k) We find the value of r by using the fact that 1. 00 mol of any gas at stp occupies 22. 4 l. Gases consist of particles constantly moving through space in random directions and with various speeds. There are no attractive forces between gas particles. The average kinetic energy (ke) of gas particles is proportional to the temperature in kelvins. Molecular collisions are elastic; when molecules collide, they may exchange ke but the total ke remains constant. Molecules collide with the walls of their container; these collisions constitute the pressure of the gas.