CHEM 1210 Lecture Notes - Lecture 6: Carbon Tetrachloride, Benzene, Physical Constant
24 views3 pages
Document Summary
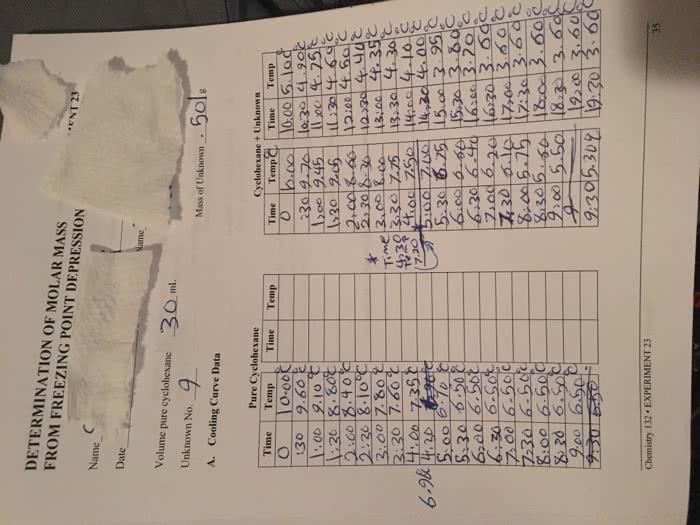
Solvent - the fraction of a solution in which the other components are dissolved. Solute - a substance that is dissolved in a solvent to produce a solution. The distribution of particles in a solution is uniform. The components of a solution do not separate on standing. A solution cannot be separated into its components by filtration. For any given solvent/solute combination, it is possible to make solutions of many different compositions. The separation is a physical change, not a chemical change. Solubility: the maximum amount of a solute that dissolves in a given amount of solvent at a given temperature. Each solid has a different solubility in every liquid. Those with low solubility are said to be insoluble, those with higher solubility are said to be soluble. Some liquids are insoluble in each other, as for example, gasoline in water (polarity: like dissolves like).
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232