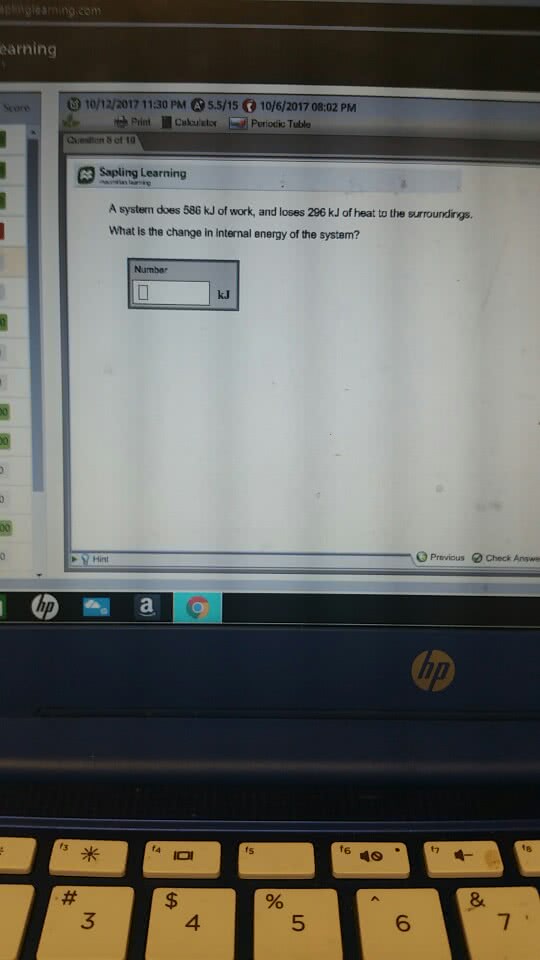
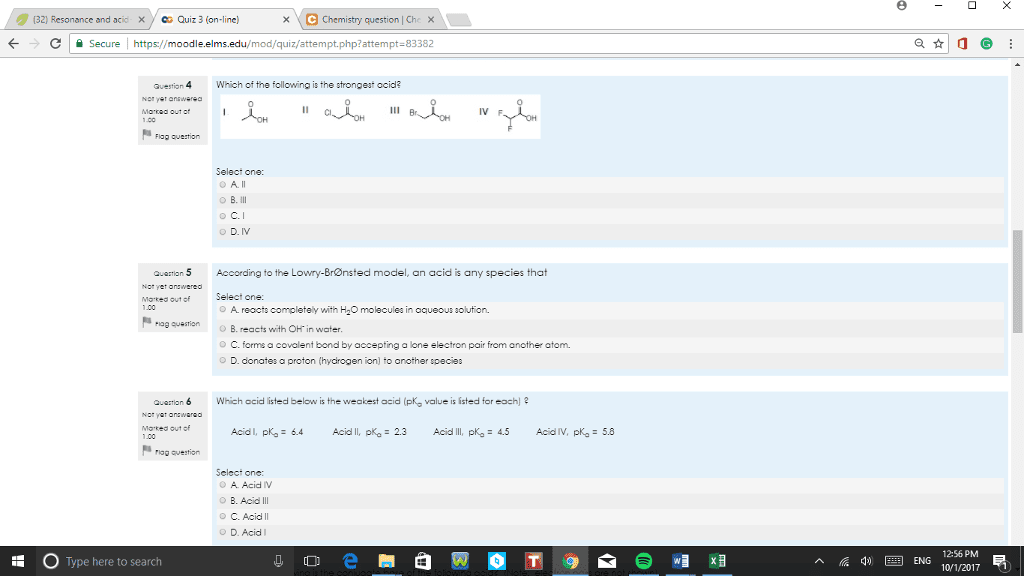
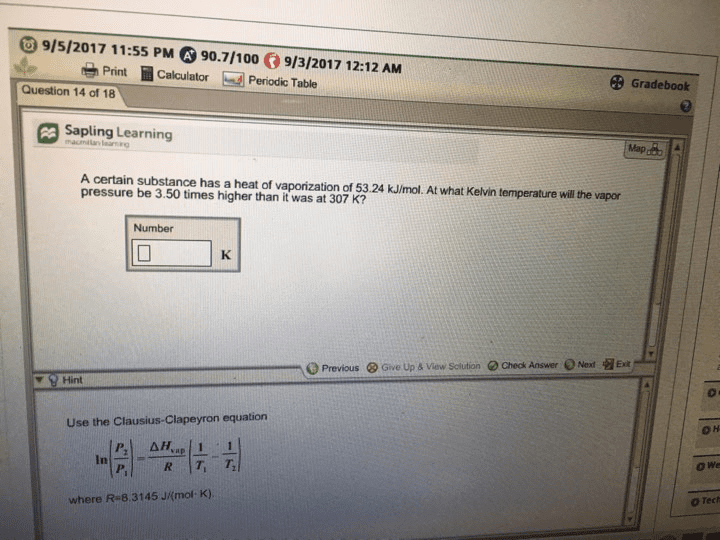
CHM 11200 Lecture 4: Lecture 4 1192017
Document Summary
Amount of energy required to increase the surface area of a liquid by a unit amount. Higher temperature leads to lower surface tension. Hydrophobic tail: dissolves oil and gets carried in the water. Resistance to flow: generally viscosity of liquid is low, depend on size and intermolecular force strength, large molecules get tangled in each other to increase viscosity, generally decreases when temperature increases. Net number of gas molecules remain constant. Partial pressure of gas in equilibrium with a liquid (in a closed container) Higher temperature ==> higher kinetic energy ==> moves into gas phase more easily. 1 atm line tells the temperature something boils at ==> normal boiling point. Change proximity and motion or particles when state changes. Identity of substance doesn"t change just the physical properties. Heat of fusion: heat required to convert a substance from solid to liquid at melting point, water = 333. 6 j/g.