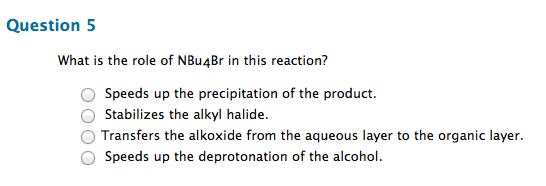CHM 25500 Lecture Notes - Lecture 33: Alkoxide, Leaving Group, Electrophile
Document Summary
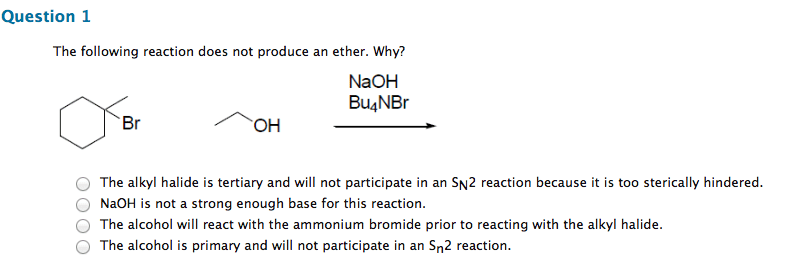
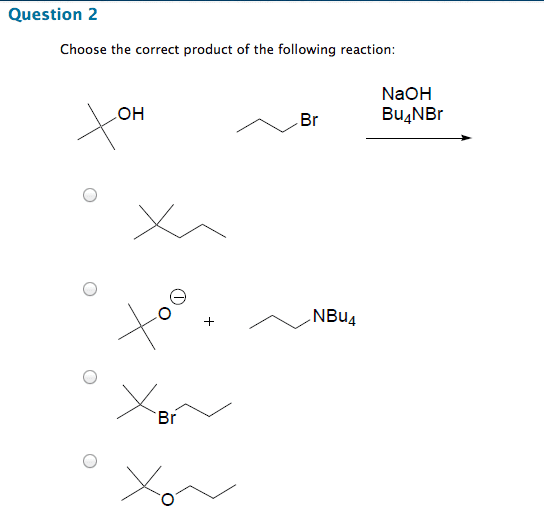
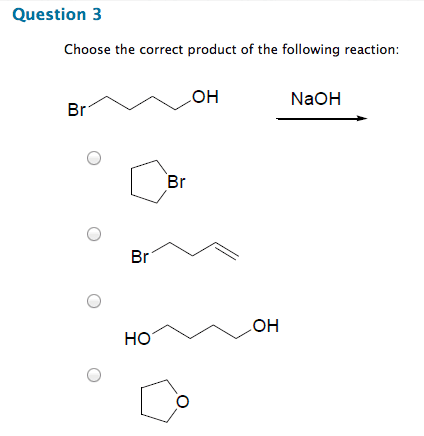
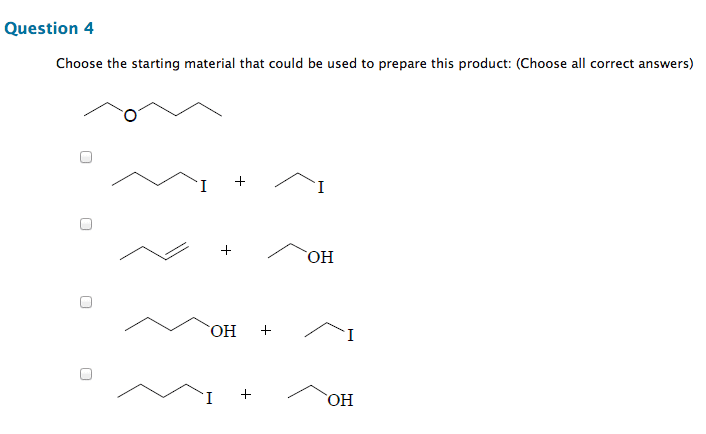
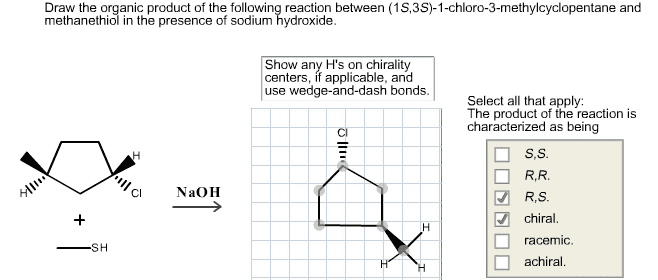
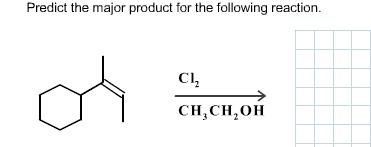
Chm 255 - lecture 33 - reactions of alcohols. An alkoxide (deprotonated alcohol) may act as a nucleophile and can attack an alkyl halide as in the following reaction. Reactions like this are called williamson ether syntheses; they involve the reaction of an alkyl halide with an alcohol to produce an ether. Hydroxide ions make for poor leaving groups, therefore, in order to behave as an electrophile, alcohols must protonated to turn the hydroxyl group into water, a much better leaving group. Dehydration of alcohols occurs in the presence of concentrated strong acids. Dehydration is the removal of a molecule of water from the alcohol (hydroxyl group + hydrogen) The preferred product is the one where the double bonded carbons are the most substituted. Oxidation involves either the addition of oxygen or the subtraction of h2. Primary alcohols may be oxidized by subtracting an equivalent of h2 to form aldehydes. Aldehydes may be further oxidized to form carboxylic acids.