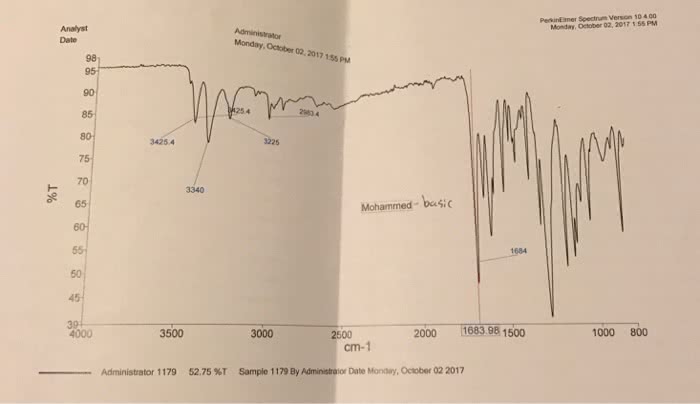
CHE 111L Lecture Notes - Lecture 9: Buffer Solution, Acetic Acid, Equivalence Point
Document Summary
The procedure for this experiment can be found on pages 54-56 in general chemistry. Carbon copies of the data table have been attached to the back of this lab report. Calculations: molarity of sodium acetate, buffer a: = 0. 327m: buffer a: ph=4. 75 + log (0. 0332m/0. 1m) = 4. 27, buffer b; ph=4. 75 + log (0. 0332m/1. 0m) = 3. 27. Buffering capacity (volume needed to change buffer solution by 2 ph units) The purpose of this experiment was to use the henderson-haselbalch equation to determine the amount of acetic acid and sodium acetate needed to prepare two acidic buffer solutions. Buffer solutions were then prepared and their buffering capacitates were tested by adding solutions of. The molarity of acetic acid was found to be 0. 1m in buffer a and 1. 0m in buffer b. The molarity of sodium acetate was found to be 0. 0332m in buffer a and 0. 327m in buffer b.