CHEM 06100 Lecture Notes - Lecture 3: Molar Mass, Unified Atomic Mass Unit, Stoichiometry
81 views1 pages
31 Oct 2017
School
Department
Course
Professor
Document Summary
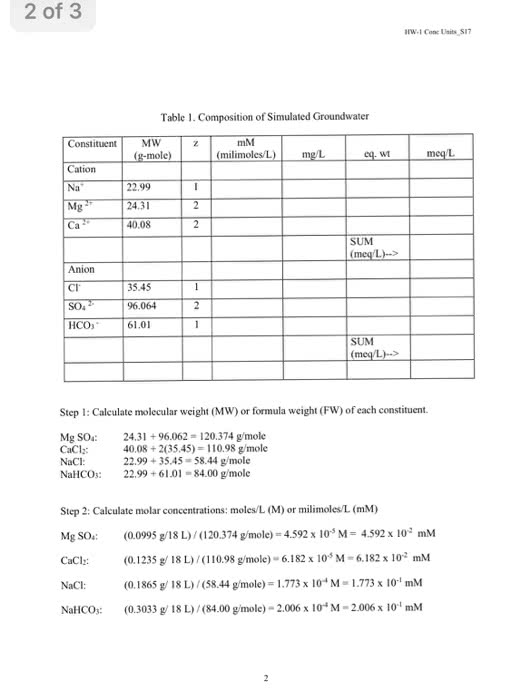
Stoichiometry and chemical reactions: stoichiometry- to determine how much mass, moles, and the making of product. The mole (mol) if we take 12 grams of carbon 12 we have 1 mol. An atom: na= 1 x m of na = 22. 99g/ mol (amu) O2= 2 x m of o = 2 x 16. 00 g/mol = 32. 00g/mol (amu) H2o= 2 m of h= 2. 00+ 1 mo= 16. 00=18. 00g/mol. An ion (particle)= ci= 1 x < =35. 45 g/mol. A formula unit nacl= 1 x m of na = 22. 99+1 x m of ci = 35. 45 g/mol = 58. 44g/mol: need a transfer electron and electronegativity to make ion. [ atoms of x in formula] x [ atomic mass of x (amu)] [moles of x in formula]x [ molar mass of x(/mol 0] Mass fraction and the mass of an element. Mass of compound x mass of element in 1 mol of compound.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232