01:160:308 Lecture Notes - Lecture 28: Hydrogenation, Electrocyclic Reaction, Stereospecificity
Document Summary
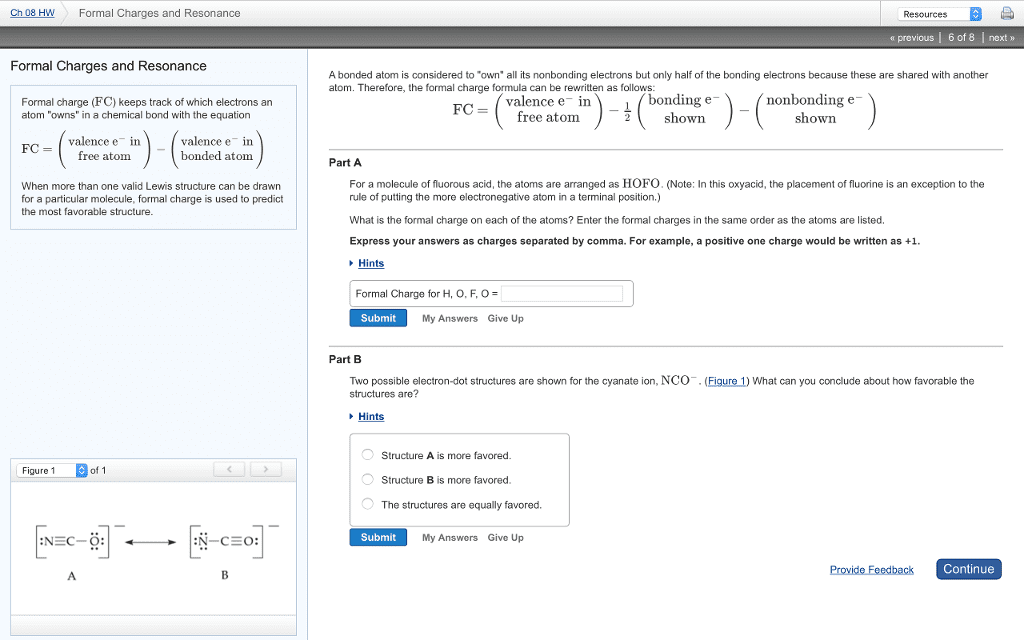
Chapter 20 conjugated dienes: delocalization and conjugation, valence bond and molecular orbital approach of conjugated systems, reaction of conjugated dienes, pericyclic reactions, uv spectroscopy, delocalization and conjugation: Molecules which can be represented by more than one lewis structure with the same placement of atoms due to different distribution of pi and nonbonded (lone pairs) of electrons are said to be in resonance. The different lewis structures are called contributing structures. The actual structure is resonance hybrid of various contributing structures, i. e. in actual structure the electrons are delocalized between more than two centers. The actual structure has all n-o bond length equal and the bond length is in between n-o single bond and n=o bond. Thus, the to e density is delocalized between bonds. Rules to draw resonating structure: all contributing structures must have same no of valence electrons, no change in atom position allowed.