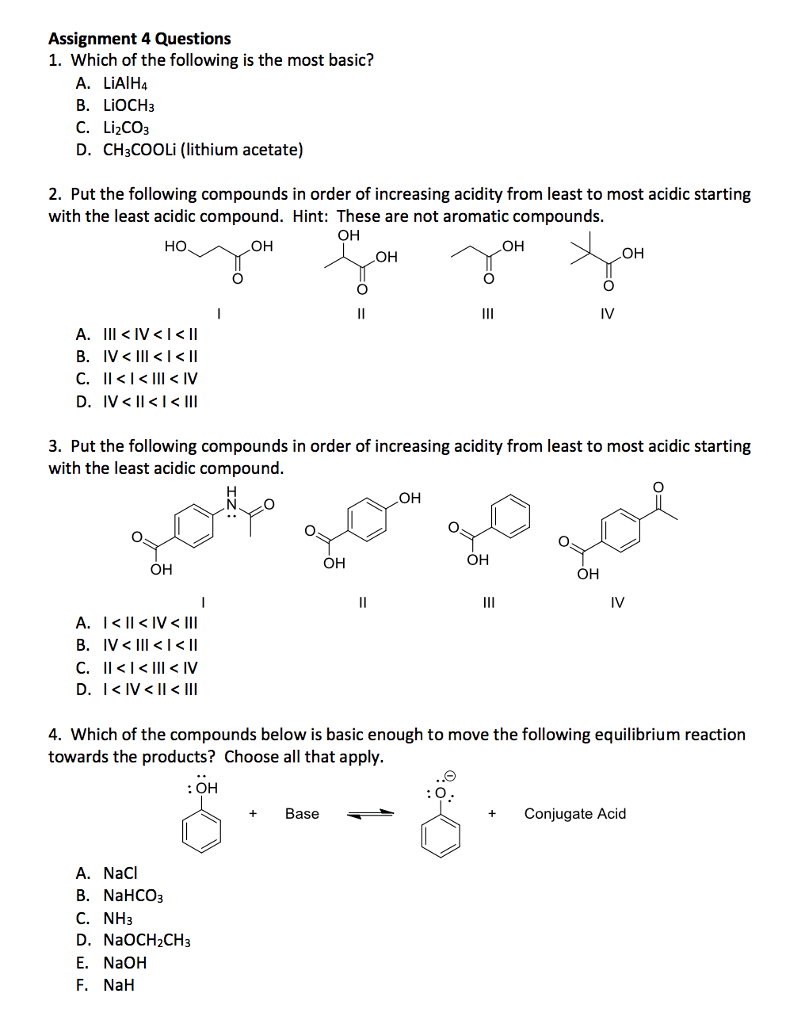
1. A 72.1 ml sample of ethyl alcohol contains14.6 grams of ethyl alcohol and 53.6 grams of water. Ethyl alcoholhas a molecular weight of 46.067 g/mol. Water has a density of 1.0g/mL.
A.) What is the mole fraction of ethyl alcohol in thissolution?
B.) What is the molality of the solution?
C.) What is the Molarity of the solution?
2. Consider intermolecular forces and arrangethe following in order of increasing boiling point. ExplainWhy.
I. CH3CH2OH
II. CH3CH2CH3
III.H3C-O-CH3
IV. CH3CH2NH2
a. IV < III < II < I
b. II < III < IV < I
c. I < IV < III < II
d. II < III < I < IV
e. III < IV < I <II
3. Commercial cold packs often contain solidNH4NO3 and a pouch of water. The temperature of the pack drops asthe NH4NO3 dissolves in water. Therefore, for the dissolving ofNH4NO3 in water,
A. delta H of solution is negative and delta S of solution maybe negative or positive
B. delta H of solution is negative and delta S of solution ispositive
C. delta H of solution is positive and delta S of solution maybe negative or positive
D. delta H of solution is positive and delta S of solution ispositive
Explain Why.