CHE 1220 Lecture Notes - Lecture 12: Rate-Determining Step, Elementary Reaction, Rate Equation
Document Summary
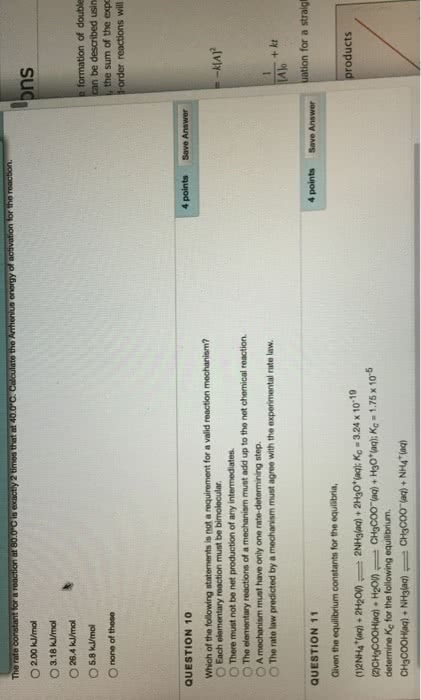
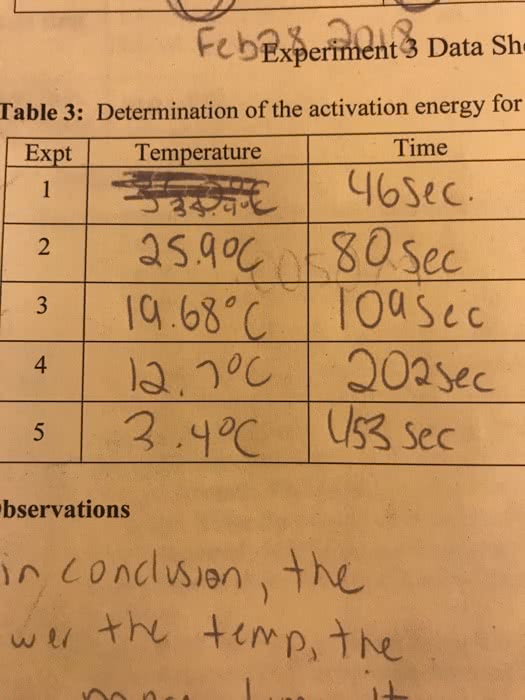
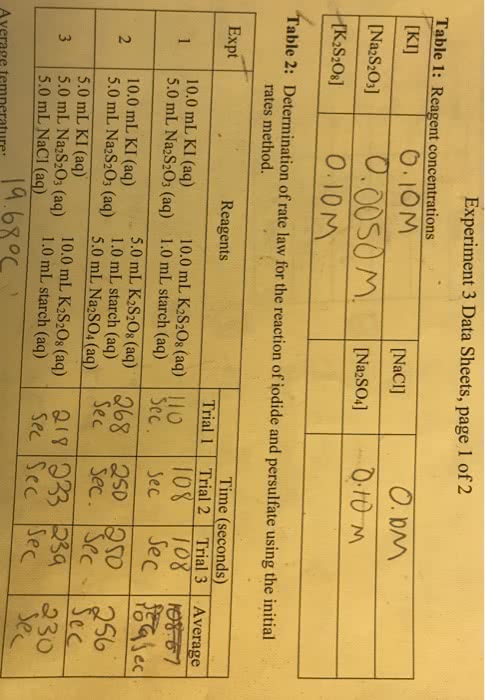
A reaction mechanism is a sequence of elementary reactions that describes what we believe takes place as the reactant molecules are transformed into product molecules. A rate law can be predicted from a proposed reaction mechanism, and you can compare predicted and experimental rate laws to determine whether a proposed reaction mechanism is consistent with the experimental rate law. In order for the mechanism to be correct, the predicted rate law must match the rate law that is determined experimentally. Not all collisions between molecules result in a reaction. The molecules must have enough energy for the reaction to occur. The minimum energy that is needed is called the activation energy. Catalysts are substances that lower the activation energy and thereby increase the rate of reactions. Catalysts are essential in the production of industrial chemicals. Biological catalysts, which are called enzymes, are essential for life and for the development of new pharmaceutical products.