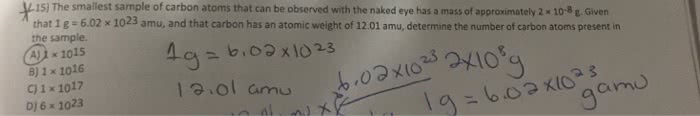CHE 106 Lecture Notes - Lecture 7: Unified Atomic Mass Unit, Molar Mass, Chemical Formula

63
CHE 106 Full Course Notes
Verified Note
63 documents
Document Summary
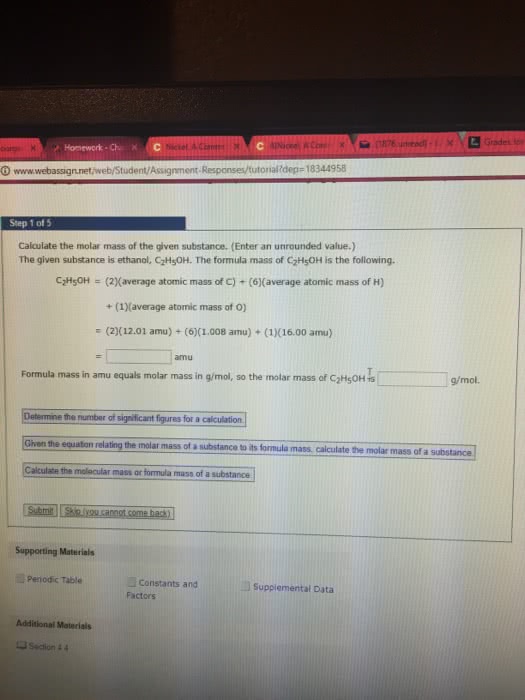
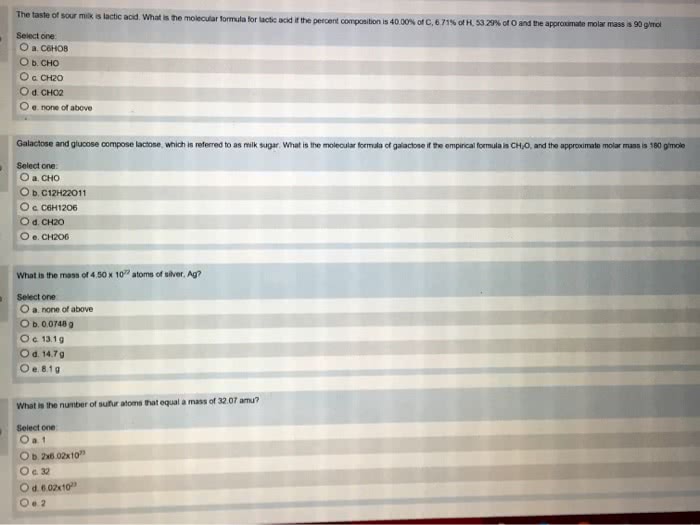
12 h x 1 g/mol = 12g h. 6 o x 16g/mol = 96 g o. 1 mole glucose = 180g: molar calculations moles. Moles fe = g/aw mole fe = 4g/55. 8 g/mol: mole fe = 0. 0717 mole fe, 2. 1 mole oxygen: empirical formulas from elemental analysis. What is the empirical formula: 100 g sample ^ 17. 2 g h^ 82. 8 g c ^ If the sample is composed of 17. 2 g hydrogen and 82. 8 grams carbon, what is the empirical formula: 1. Need to convert grams to moles: h 17. 2g / 1g/mol = 17. 2 mole h, c 82. 8g / 12g/mol = 6. 9 mole c, 2. Need to find mole ratio: 17. 2 mol h / 6. 9 = 2. 49 mol h, 6. 9 / 6. 9 = 1 mole c, c6. 9 h17. 2, for the purposes of this calculation, assume we have 100. 0 grams of benzoic acid. Remember: the compound contains 68. 8% c, 5. 0% h, and 26. 2% o by mass.