Please provide mechanism for this reaction
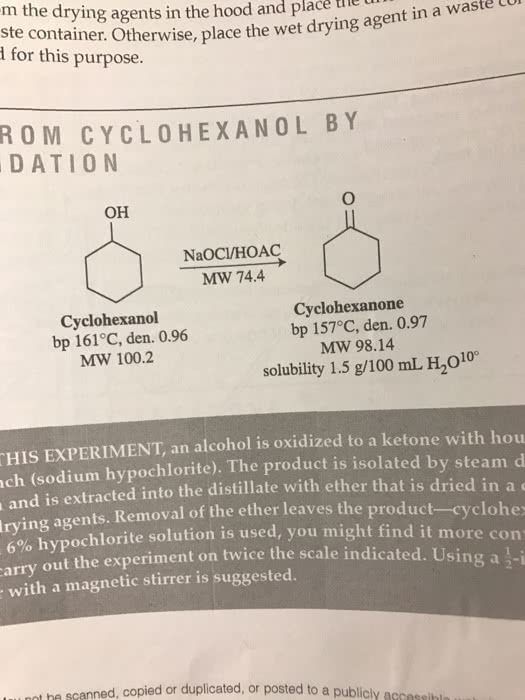
m the drying agents in the UUl and drying agent in a ste Otherwise, place the for this purpose. M CY CLO HEX A N OL B Y TION OH NaOCl/HOAC MW 74.4 Cyclohexanone Cyclohexanol bp 157°C, den. 0.97 bp 161°C, den. 0.96 MW 98.14 MW 100.2 solubility 1.5 g/100 mL 10° HIS alcohol is oxidized to a ketone with hou EXPERIMENT, an d (sodium hypochlorite). The product is isolated by steam and is extracted into the distillate with ether that is dried in a drying agents. Removal of the ether leaves the product-cyclohex 6% hypochlorite solution is used, you might find it more con arry out the experiment on t the scale indicated. Using a t-i with a magnetic stirrer is suggested. not nn scanned, copied or duplicated, or posted to a publicly accese















