Solution Investigation Question: What are some of the key characteristics of unknown sample of an aqueous solution containing an ionic compound?
1. Given that the cation is from Group 1A, what is the identity of the group 1A metal ion in your solution?
2. Given that the ionic compound contains either chloride or hydroxide, how can you determine which? (Two pieces of evidence are required and one must be related to solubility rules)
3. What is the concentration of your solution? (Your unknown sample is known to be about 1 M, but you need to determine the concentration to 3 significant figures using titration. You should plan ahead so that you know approximately what volume of your sample you will need in order to use approximately 25 mL of 0.100 M sulfuric acid for the titration).


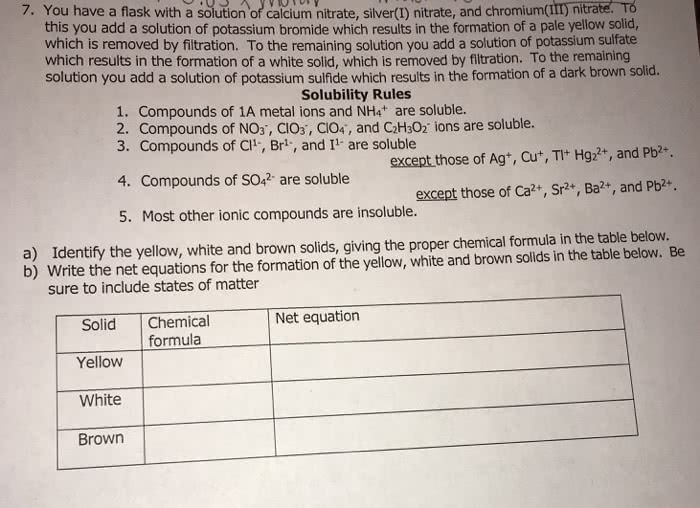
Resources Solubility Rules for Salts (lonic Compounds) Soluble Compounds Insoluble Compounds o Compounds of Group 1 elements o Carbonates (CO2), chromates (Cror2), Ammonium (NH4.)compounds o Chlorides (Cl), bromides (Br), and iodides oxalates (COr), and phosphates (POr), except those of the Group 1 elements and (I), except those of Ag, Hg2 and Pb NH o Nitrates (NOs), nitrites (NO:), acetates o Sulfides (S), except those of the Group 1 (CH CO2), chlorates (CIOs), and perchlorates (C1O:) and 2 elements and NH4. Hydroxides (OH) and oxides (O2), except those of the Group 1 and 2 elements, and NH o o Sulfates (SOr2), except those of Sr, Ba Pb2 and Hg