CHEM 1A Lecture Notes - Spin Quantum Number, Wave Function

44
CHEM 1A Full Course Notes
Verified Note
44 documents
Document Summary
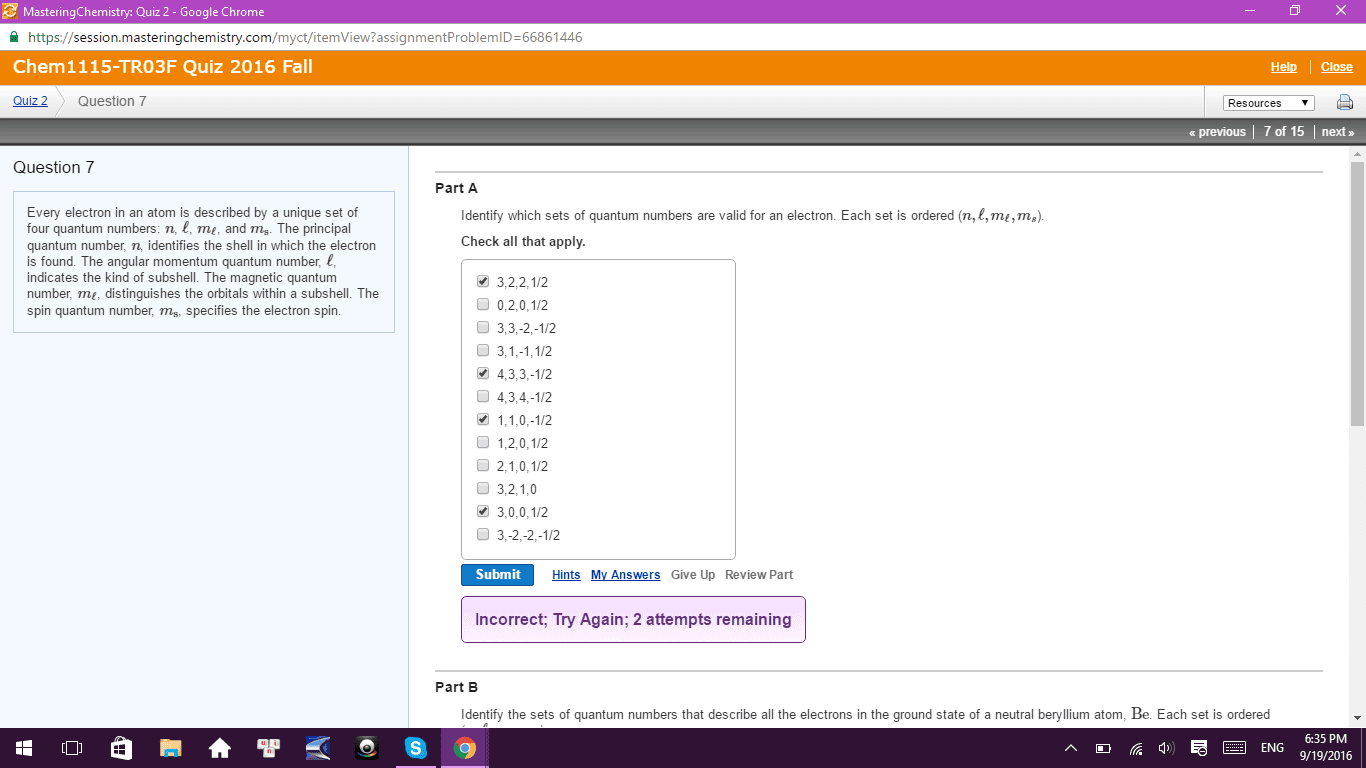
The combination of them all specifies the wavefunction. You can think of it as an address to an electron. Building number 403 room number 1100 row fchair 1. Quantum numbers n l m m s l. We saw this in the ____________ as the energy ?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)?(cid:0)? (cid:0) These are associated with the atomic orbitals we just discussed. Can be any integer from 0 to n-1. Question: for the 4th energy shell, what are the orbitals present? n. Quantum numbers- magnetic quantum number magnetic quantum number- ml (read as m sub l) Distinguishes orbitals that have same n and l (same energy and shape) but having a different orientation. Table of allowed values for 1-4 energy levels m l n l. Lets fill in the numbers for the n=3 energy level. l=0 s ml=0 l=1 p ml= -1 0 1 l=2d ml=2 -1 0 1 2.