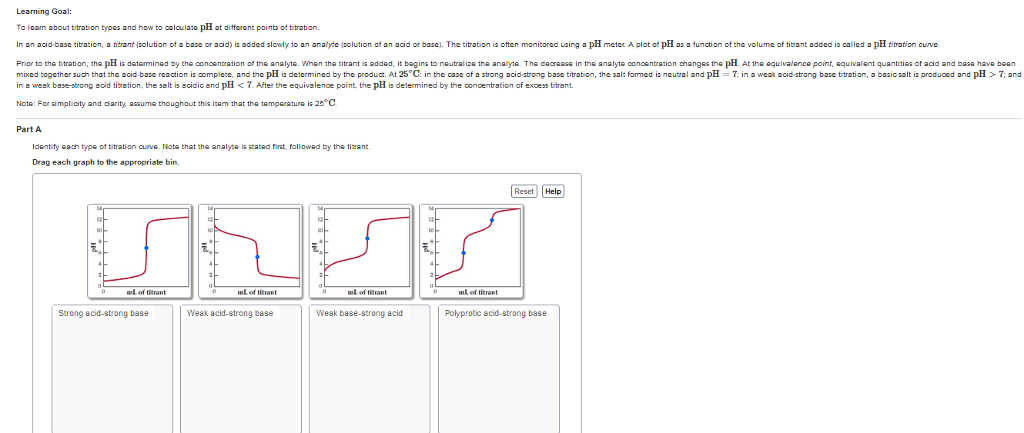
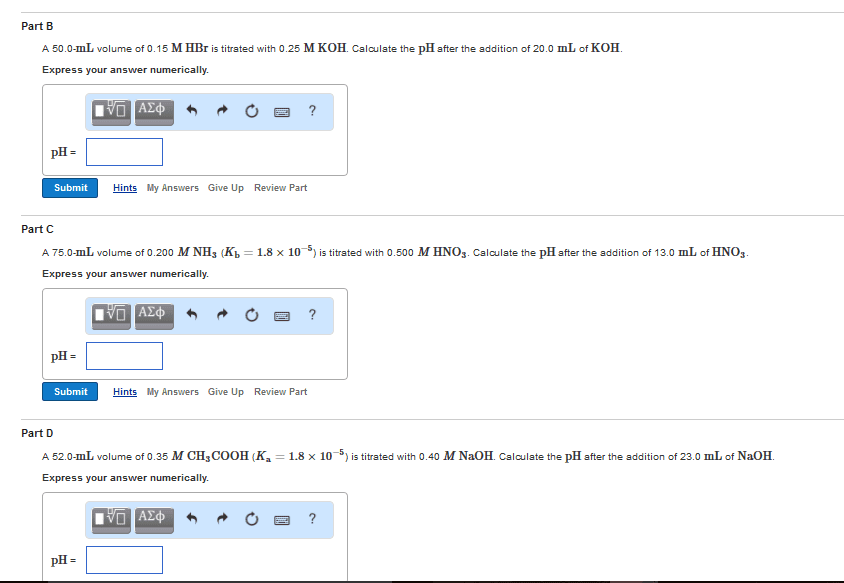
Learning Goal To learn about titration types and how to alculata pH at ditterent paints of titration. In an acid-base titration, a titrant zolution of base or acid) i added slowly to an analyte (solution of an acid or basc). The titration is often monitored using a pH meter A plot of pH as a function of the volume of titrant added called o pH titration curve. is Prior to the titration, the pH is determined by the concentration of the analyte. When the titrant is added. it begins to neutralize the analyte. The dearease in the analyte oonoentration changes the pH At the equivalence point, equivalent quantities of acid and base have been mixed together such that the d-base reaction is oomplete, and the pH is determined by the product. At 25 C: in the case of a strong strong base titration, the salt formed is ncutral and pH n o weak strong base titration, basic salt is produced and pH 7; ond in a weak base-strong acid titration, the salt is acidic and pH



