CHEM 20A Lecture 3: [Wk1.Wed] Moles and Molar Mass
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
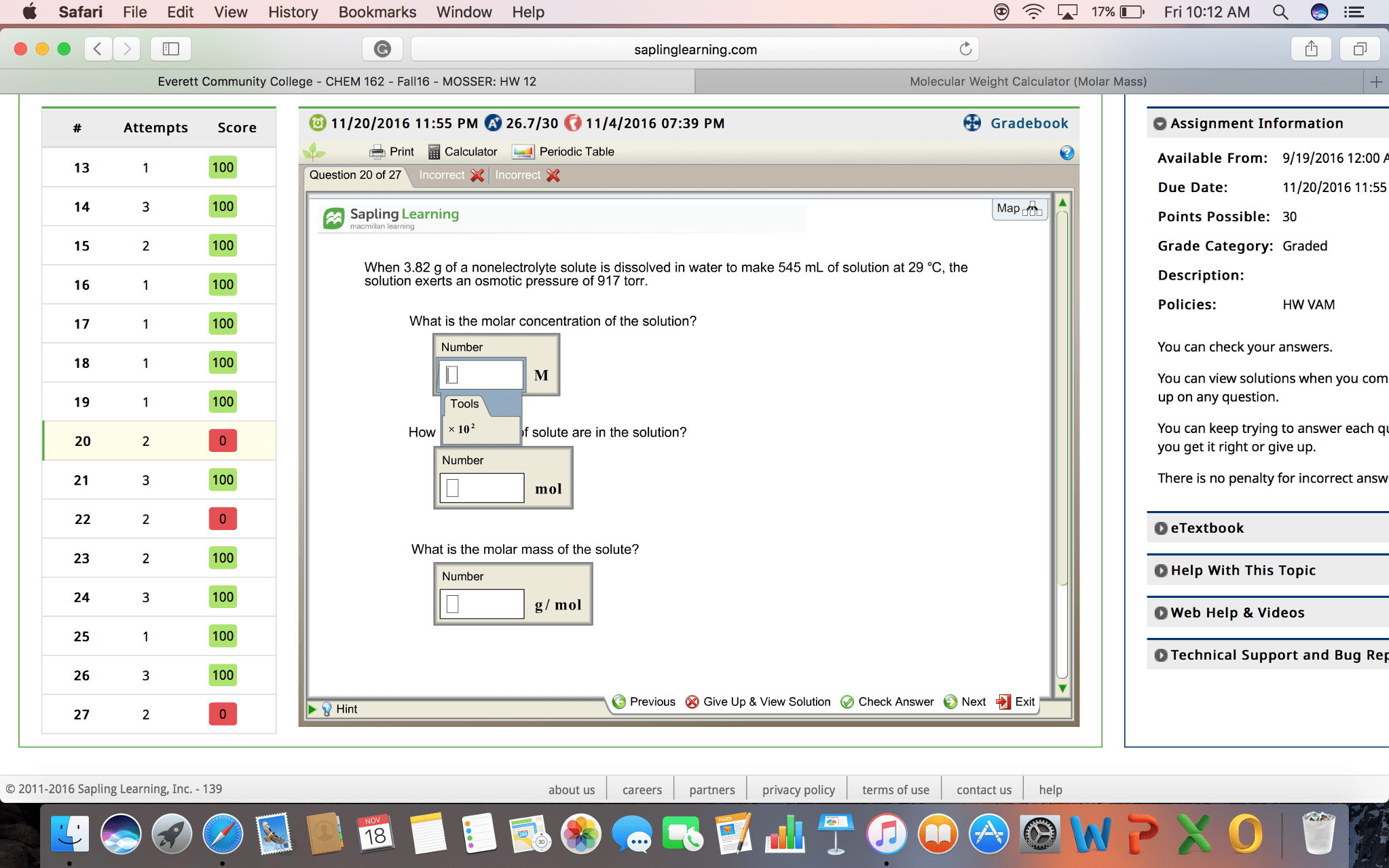
Exercise 1: Determining the Concentration of Acetic Acid
Data Table 1. NaOH Titration Volume.
| Initial NaOH Volume (mL) | Final NaOH Volume (mL) | Total volume of NaOH used (mL) | |
| Trial 1 | 9 | .5 | 8.5 |
| Trial 2 | 9 | .25 | 8.25 |
| Trial 3 | 9 | 0 | 9 |
| Average Volume of NaOH Used (mL) : 8.58 | |||
Data Table 2. Concentration of CH3COOH in Vinegar.
| Average volume of NaOH used (mL) | Concentration CH3COOH in vinegar (mol/L) | % CH3COOH in vinegar |
* I don't know if I am doing it correctly, and end up with crazy numbers. Can someone PLEASE break it down easily for me?
Question
The manufacturer of the vinegar used in the experiment stated that the vinegar contained 5.0% acetic acid. What is the percent error between your result and the manufacturerâs statement?
UPDATE: NaOH is 0.5M
I DO NOT know CH3COOH.... It was for me to find out in that chart.....
UPDATE AGAIN: I know he acedic acid was not given. That was suppose to be what I am figuring out. This is what it says for data:
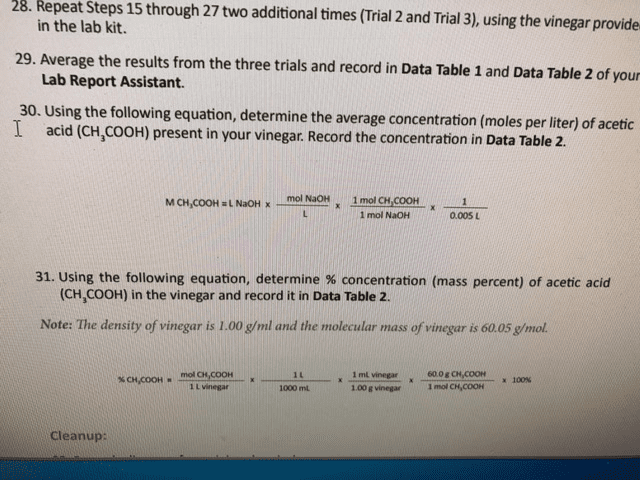
UPDATE AGAIN: I DO NOT KNOW THE VOLUME BECAUSE THAT IS WHAT IS IN THE BLANKS THAT I NEED TO FIGURE OUT!!!!!!!!!!!