CHEM 1104 Lecture Notes - Lecture 6: Atomic Orbital, Radiant Energy, Photon
36 views3 pages
Document Summary
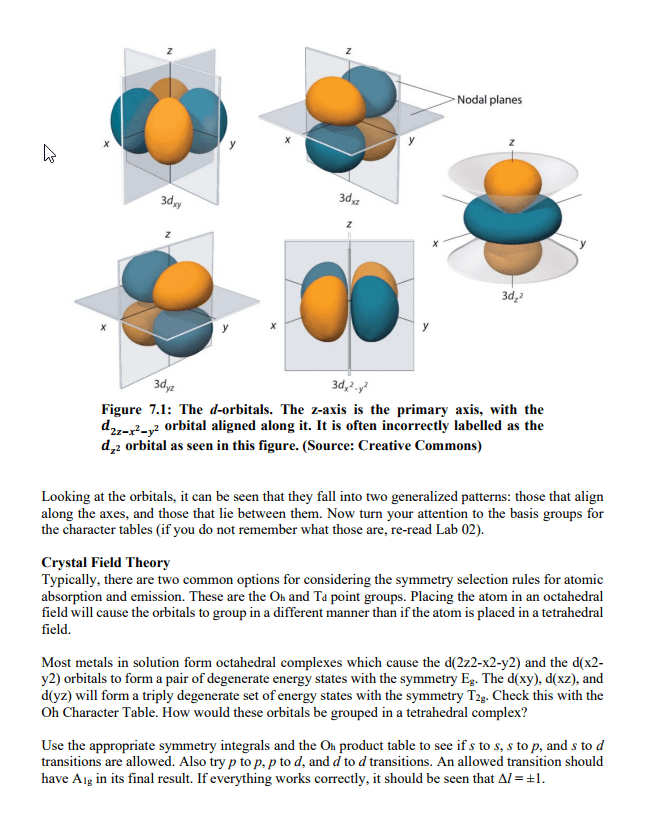
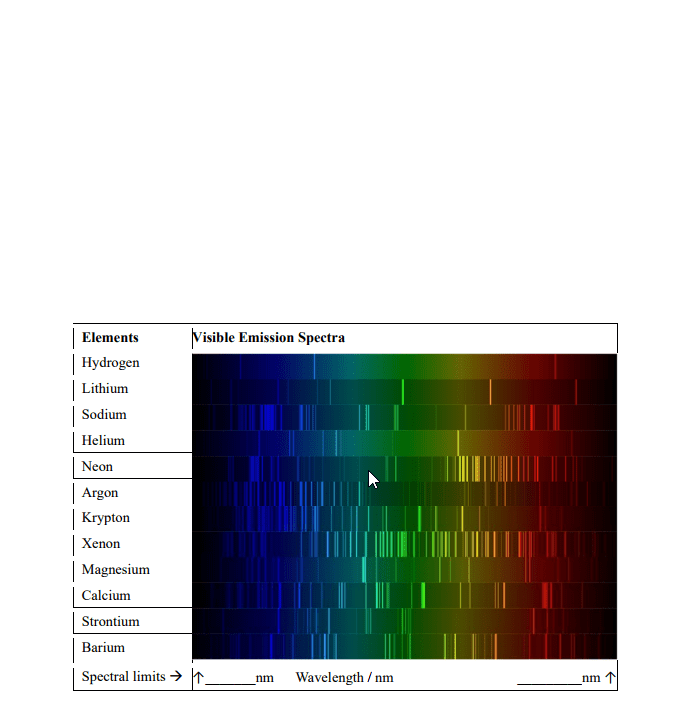
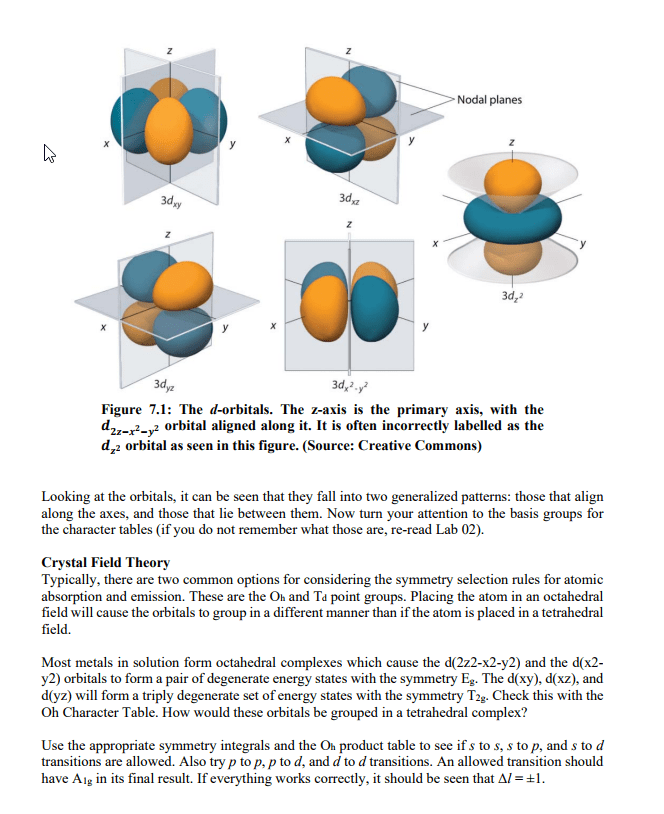
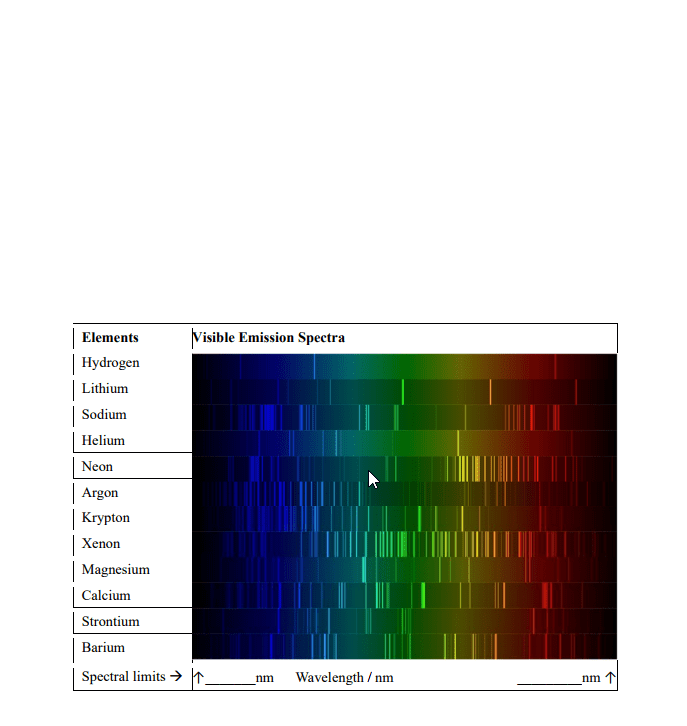
Frequency= indicates how many wave peaks pass a certain point per given time period. Speed= how fast one particular wave travels, through a space. An atom is said to be in an excited state when it has excess energy. When an atom is in the lowest possible energy state; it in the ground state. The energy of the photon corresponds to the change in energy that the atom experiences. All hydrogen atoms have the same set of quantized levels. Quantized energy levels result in emission of only certain colors. Has to go from one state to another; no in between. The energy levels of all atoms are quantized. Electron jumps between levels by absorbing or emitting a photon of a particular wavelength. Involves orbitals= regions where there is a high probability of finding an electron. One can predict the probability of finding the electron at given. An orbital"s size is the sphere that contains 90% of the total electron probability.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232