CHEM103 Lecture Notes - Lecture 13: Formal Charge, Octet Rule, Boron
Document Summary
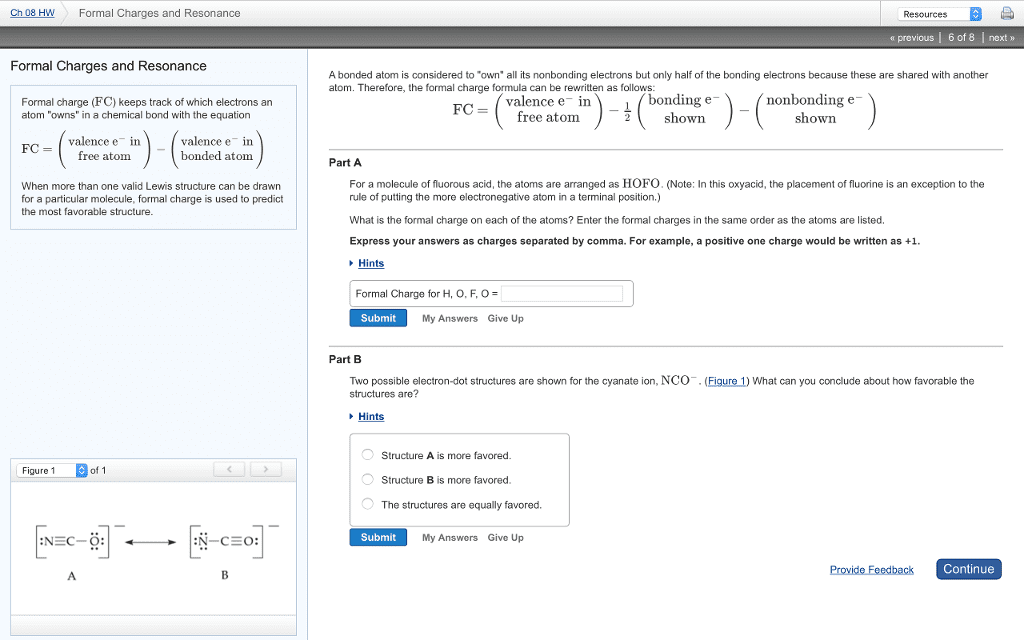
Chapter 9 notes: charge an atom would have if all bonding electrons were shared equally between bonded atoms. Fc=valence e- (-) nonbonding e- (-) bonding e- Sun of all the formal charges in a molecule=0: evaluating resonance structures. Better structures have the negative formal charge on the electronegative. Exception to the octet rule atom: boron does not need 8, it is happy with 6 electrons. Way for boron to get full octet: form an adduct: expanded octet. Each lone pair of electrons constitutes one electron group on a central atom. Each bond constitutes one electron group on a central atom, regardless of whether it is a single, double, or triple bond. November 7, 2016: another way to look at it. Count the number of atoms directly connected to the central atom. Count the number of lone pairs on the central atom: 2 electron groups: linear. 2 electron groups around a central atom.