CHM 2045 Lecture Notes - Rate Equation, Reaction Rate, Reaction Rate Constant
Document Summary
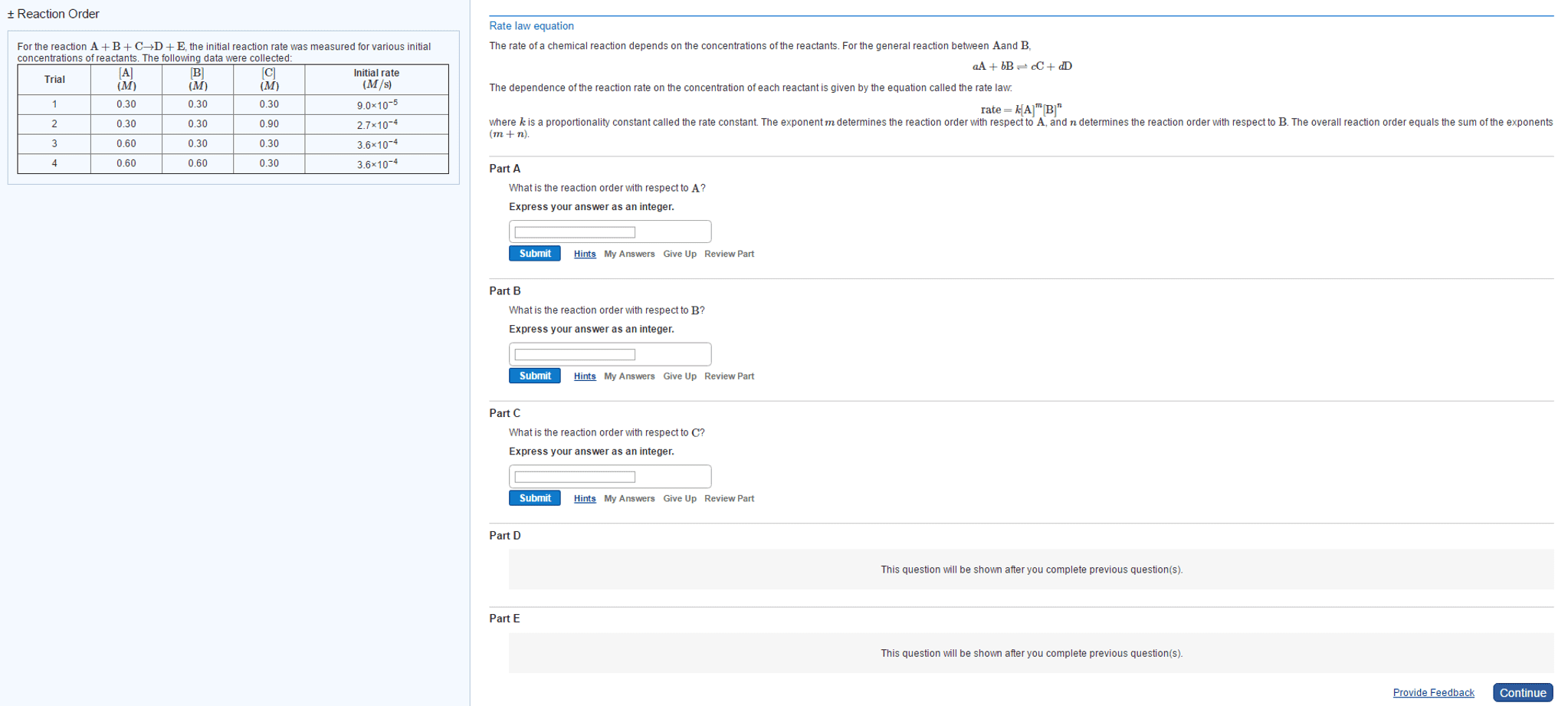
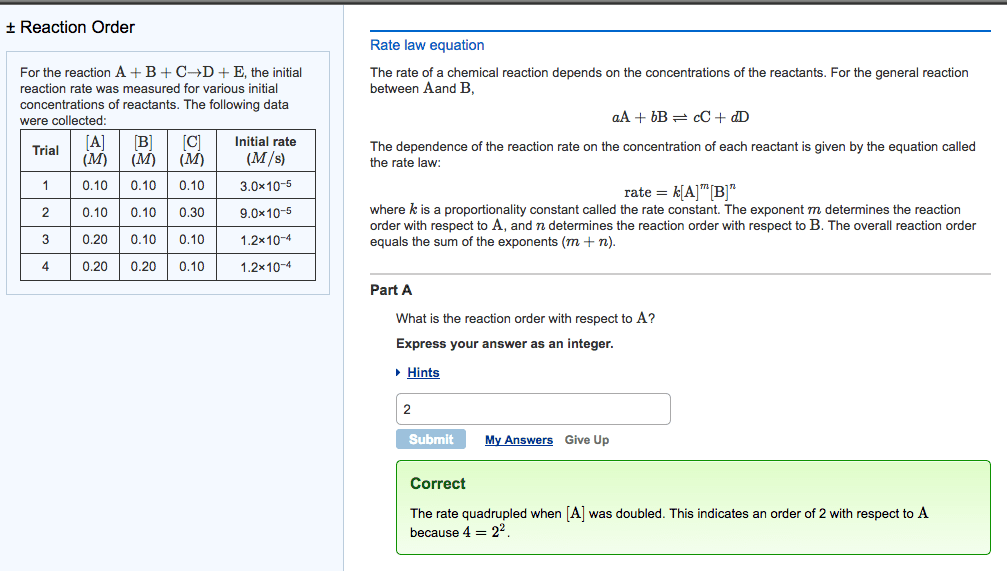
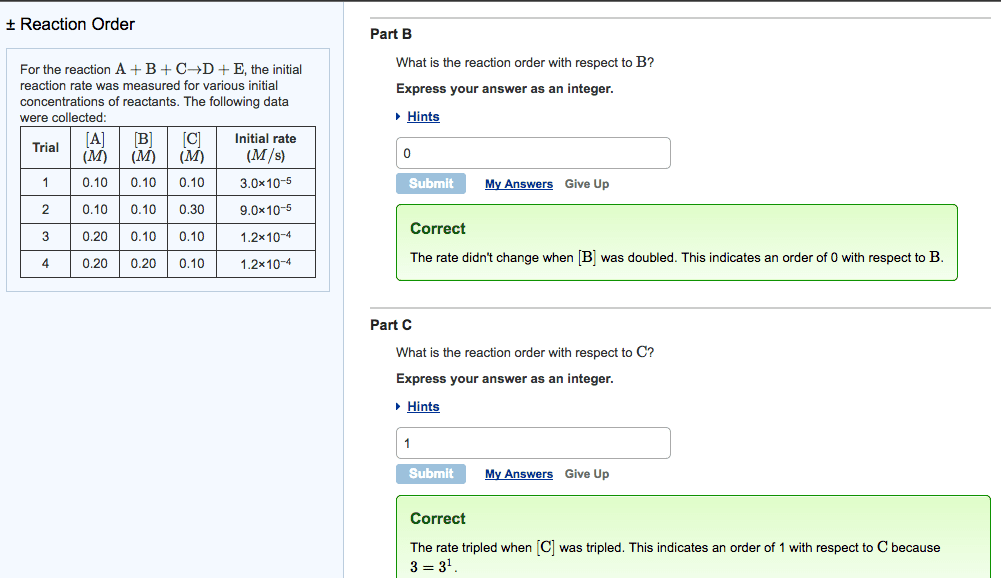
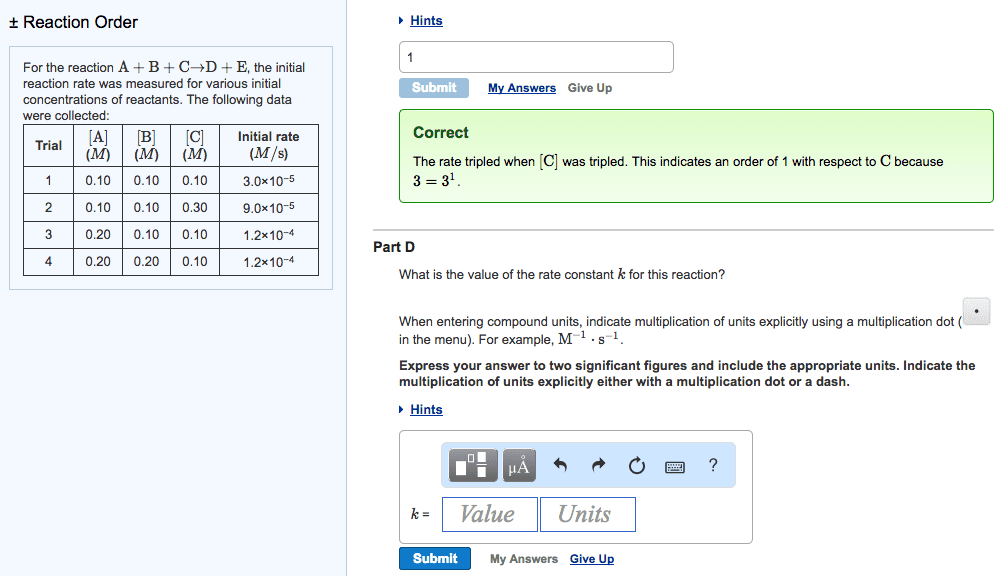
Rate of a reaction says how quickly [product] or [reactant] is changing: b. Given in molarity per second, mol/l*s: c. factors affecting reaction rates, temperature, pressure usually negligible, [substances, reaction mechanisms, reaction order. Reaction order = sum of all exponents of the concentration variables in the rate law: b. Reaction order in a = the exponent of [a: a. The k in the rate law is the rate constant: b. The rate constant is an empirically determined value that changes with different: c. within a given equation, independent of: reactions and reaction conditions, dependent on, [product, [reactant, temperature, catalyst, a. The equation that describes the rate = the product of reactants raised to some. The balance of products and reactants at equilibrium. 11: reaction rate equations: example, reaction rate equations: types balanced equation serve as the exponent in the rate law, a. aa + bb cc + dd, b.